Team:UChicago/Composite Part

***Note: Several parts that have no BBa plasmid were not submitted. We have several of these plasmids not in the registry on hand, but we forgot to put them onto a chloramphenicol backbone for submission.
This page includes all our parts we made during the summer, which includes several we weren’t able to submit. The two actual Biobrick parts submitted to the Registry are labeled.
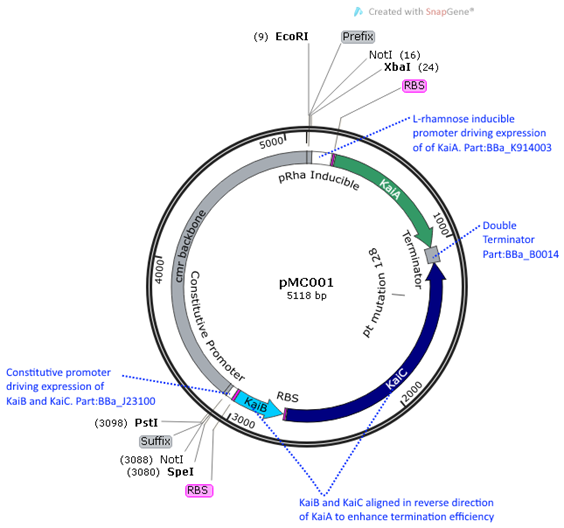 pMC001 (BBa_K1745001) is the main Kai ABC oscillator biobrick we submitted into the
iGEM registry. It contains the three Kai proteins -KaiA, KaiB, and KaiC under
the expression of two different promoters. KaiB and KaiC are under a
constitutive promoter (Part J23100) and KaiA is under a L-rhamnose inducible
promoter (Part K914003). The inducible promoter was chosen for KaiA in order to
investigate the effect of altering the stoichiometric ratios between KaiA and
KaiC. This ratio was found to be particularly significant in in vitro
reactions, where the ratio of KaiA to KaiC was 1:3 (Nakajima, Science 2015).
pMC001 (BBa_K1745001) is the main Kai ABC oscillator biobrick we submitted into the
iGEM registry. It contains the three Kai proteins -KaiA, KaiB, and KaiC under
the expression of two different promoters. KaiB and KaiC are under a
constitutive promoter (Part J23100) and KaiA is under a L-rhamnose inducible
promoter (Part K914003). The inducible promoter was chosen for KaiA in order to
investigate the effect of altering the stoichiometric ratios between KaiA and
KaiC. This ratio was found to be particularly significant in in vitro
reactions, where the ratio of KaiA to KaiC was 1:3 (Nakajima, Science 2015).
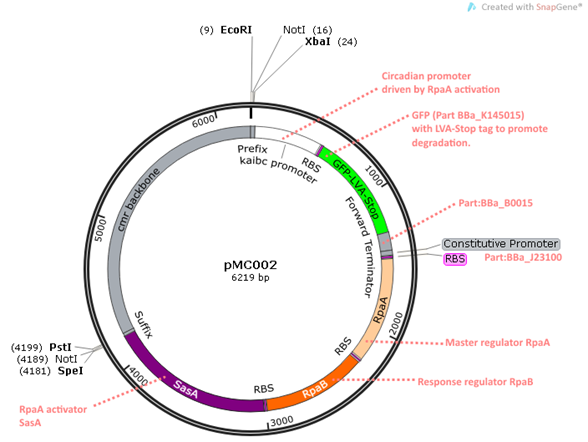
pMC002 is one of the constructs for the read-out system. It contains the circadian promoter kaibc which is driven by the master transcription factor RpaA. RpaB is included as some studies have reported both RpaA and RpaB necessary for kaibc expression (http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3406716/ Hanoaka, 2012). This was also to reconstitute the system used by the Silver lab. SasA, the RpaA activator is included as it directly binds to KaiC and phosphorylates RpaA, linking the KaiABC oscillator to an expression based read-out system. GFP is placed downstream the circadian promoter, acting as a reporter for kaibc expression. The GFP is LVA-Stop tagged in order to promote degradation, which is useful to quantify increases and decreases of GFP which we expect to see throughout the KaiC oscillations.
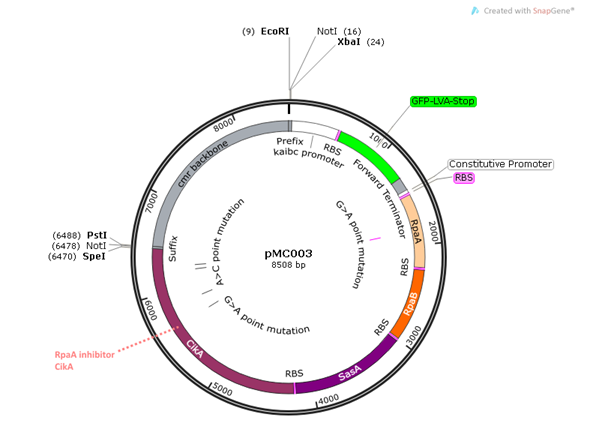
pMC003 is another read-out plasmid that will be assayed independently of pMC002. This construct exhibits the same features as pMC002 with the addition of CikA, a negative regulator of RpaA which interacts directly with KaiB and dephosphorylates RpaA. We wanted to directly examine the effects of the read-out system with and without the CikA negative regulator to test the hypothesis that the inclusion of CikA leads to more robust regulation of RpaA.
Both pMC002 and pMC003 will be transformed in vitro with pMC004-7,
representing the different phosphomimetics of KaiC.
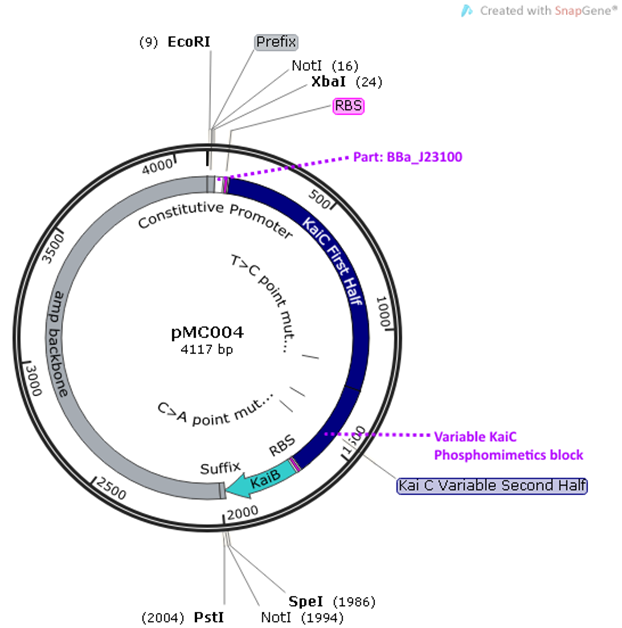 pMC004-7 are supplemental plasmids to pMC002
and 3. They contain KaiB and KaiC phosphomimetics. The phosphomimetics
represent different phosphorylation states of KaiC and emulate the oscillations
of KaiC. KaiB is included as it interacts directly with CikA, the RpaA negative
regulator. The phosphomimetics were generated by altering the amino acids at
position 431 and 432: AA (unphosphorylated) (pMC004), AE (pMC005),
EA (partially phosphorylated) (pMC006), EE (fully phosphorylated)
(pMC007).
pMC004-7 are supplemental plasmids to pMC002
and 3. They contain KaiB and KaiC phosphomimetics. The phosphomimetics
represent different phosphorylation states of KaiC and emulate the oscillations
of KaiC. KaiB is included as it interacts directly with CikA, the RpaA negative
regulator. The phosphomimetics were generated by altering the amino acids at
position 431 and 432: AA (unphosphorylated) (pMC004), AE (pMC005),
EA (partially phosphorylated) (pMC006), EE (fully phosphorylated)
(pMC007).
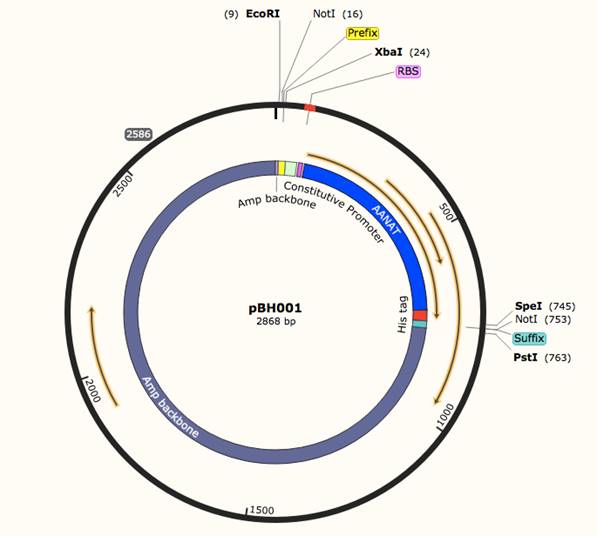 pBH001 (BBa_K1745002 ) functions in the expression and
possibly secretion of enzyme AANAT (aralkylamine N-acetyltransferase) (human).
AANAT catalyzes Serotonin into N-acetylserotonin, a Melatonin precursor, and it
is the rate-limiting step in Melatonin production. (Zheng 2002). The construct
is under a constitutive promoter (BBa_J23100) also this construct is His-tagged.
pBH001 (BBa_K1745002 ) functions in the expression and
possibly secretion of enzyme AANAT (aralkylamine N-acetyltransferase) (human).
AANAT catalyzes Serotonin into N-acetylserotonin, a Melatonin precursor, and it
is the rate-limiting step in Melatonin production. (Zheng 2002). The construct
is under a constitutive promoter (BBa_J23100) also this construct is His-tagged.
One thing that was uncertain was whether the AANAT could be secreted after expression, and if it could be secreted, whether it could retain its activity or not. We had thought about adding secretion pathway signals such as the Tat and Sec signal to the construct, and we had also thought about using addition methods such as adding phage shock protein genes and Kil genes, but in the end we decided against it because it would be hard to assay. We resolved to put these other genes on separate constructs (BH002-4)
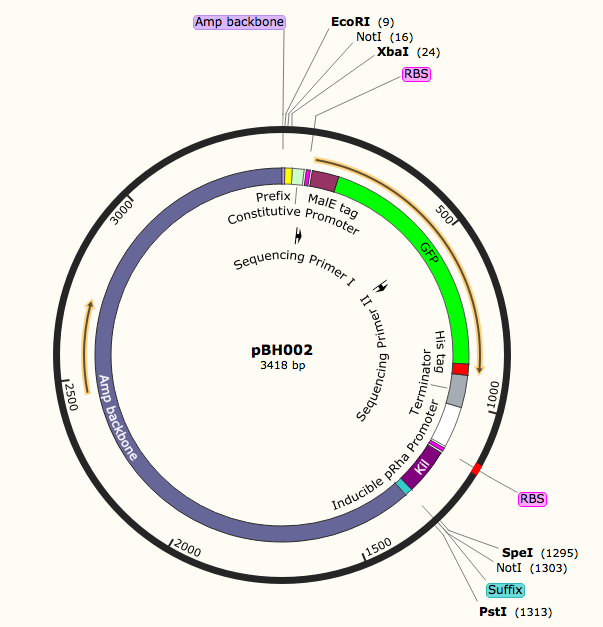 pBH002,4: These are the secretion pathway
constructs. The aim of these two constructs is to allow us to analyze
alternative methods for secretion in the case that AANAT could not be secreted
in construct pBH001. Both constructs have GFP, which we will use as a proxy for
AANAT to determine the secretion efficiency; they both have a his-tag as a back
up to GFP, and both constructs uses a constitutive promoter (BBa_J23100). pBH002 increases the expression
of the endogenous MalE tag, which is the tag for the Sec, or general secretion
secretion pathway while pBH004 increases the expression of a modified TorA tag,
which is the tag for the Tat pathway (Natale 2018). The modified TorA tag (BBa_K97002) was made by Kyoto 2012. At the end of both the constructs there
is a Kil gene coding sequence under an inducible promoter (BBa_K914003). The Kil gene leads to holes in the outer membrane of
the ecoli, which increases secretion efficiently, and it was classified by
Kyoto 2012 as well.
pBH002,4: These are the secretion pathway
constructs. The aim of these two constructs is to allow us to analyze
alternative methods for secretion in the case that AANAT could not be secreted
in construct pBH001. Both constructs have GFP, which we will use as a proxy for
AANAT to determine the secretion efficiency; they both have a his-tag as a back
up to GFP, and both constructs uses a constitutive promoter (BBa_J23100). pBH002 increases the expression
of the endogenous MalE tag, which is the tag for the Sec, or general secretion
secretion pathway while pBH004 increases the expression of a modified TorA tag,
which is the tag for the Tat pathway (Natale 2018). The modified TorA tag (BBa_K97002) was made by Kyoto 2012. At the end of both the constructs there
is a Kil gene coding sequence under an inducible promoter (BBa_K914003). The Kil gene leads to holes in the outer membrane of
the ecoli, which increases secretion efficiently, and it was classified by
Kyoto 2012 as well.
![]()
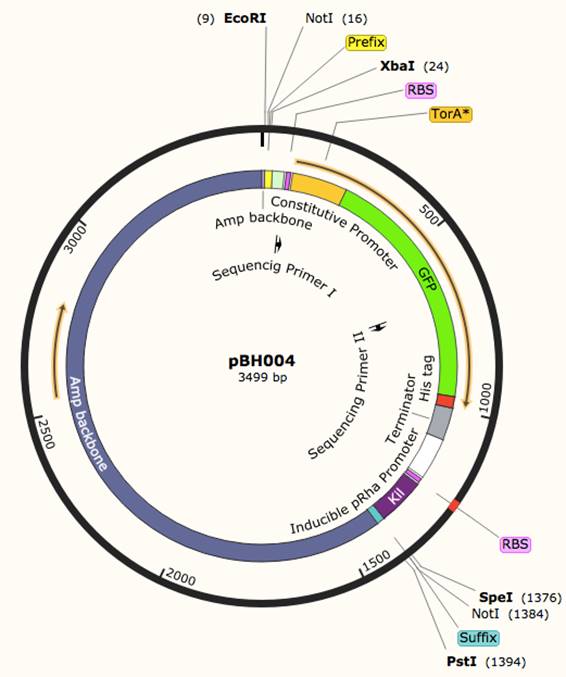
pBH003: This construct is similar to the Tat cassette developed by Kyoto 2012 (BBa_K797004 ), excepted the Kil gene is removed and placed on constructs pBH002 and pBH0024. This construct allows us to optimize the Tat secretion pathway as a possible secretion pathway for AANAT.
Like our team Facebook page, Genehackers@UChicago!
Questions? Comments? Send us an email!
