Difference between revisions of "Team:Paris Saclay/Notebook/August/10"
m |
m (→Quantification) |
||
| Line 126: | Line 126: | ||
Agarose gel 1%, migration at 100V | Agarose gel 1%, migration at 100V | ||
[[File:Paris Saclay-10.08.2015 - Quantification 2.jpg|300px|center]] | [[File:Paris Saclay-10.08.2015 - Quantification 2.jpg|300px|center]] | ||
| − | <html><p><i>Quantification, from left to right: 1. <a href="https://2015.igem.org/File:Paris_Saclay-Ladder.jpg" target="_blank">DNA Ladder</a>, 2. BBa_K1707004#1, 3. BBa_K1707004#2, 4. BBa_K1707006, 5. BBa_I13602, 6. BBa_S03518 7. BBa_R0040, 8. BBa_R0051, 9. Empty, 10. Empty, 11. Empty, 12. Empty</i></p></html> | + | <html><p><i>Quantification, from left to right: 1. <a href="https://2015.igem.org/File:Paris_Saclay-Ladder.jpg" target="_blank">DNA Ladder</a>, 2. BBa_K1707004#1, 3. BBa_K1707004#2, 4. BBa_K1707006, 5. BBa_I13602, 6. BBa_S03518, 7. BBa_R0040, 8. BBa_R0051, 9. Empty, 10. Empty, 11. Empty, 12. Empty</i></p></html> |
We can conclude: | We can conclude: | ||
| Line 135: | Line 135: | ||
* BBa_I13602 #1: 50µg/µL | * BBa_I13602 #1: 50µg/µL | ||
* BBa_R0051 #1: we can't see anything | * BBa_R0051 #1: we can't see anything | ||
| − | |||
===New Culture=== | ===New Culture=== | ||
Latest revision as of 19:59, 18 September 2015
Contents
Monday 10th August
Lab Work
Plasmid extraction
by Pauline
Biobricks:
- BBa_R0051 #1
With Macherey-Nagel Extraction kit
Digestion verification
by Coralie
Biobricks:
- BBa_K1707004 #2
- BBa_K1707011 #1
- BBa_ROO407#1
- BBa_K1707006 #1
- BBa_I13602 #1
- BBa_K1707012 #1
- BBa_R0051 #1
- BBa_S03518 #1
- Mix for BBa_S03518 and BBa_K1707006:
- 10 µL plasmid
- 1 µL XbaI
- 1 µL PstI
- 2 µL Buffer FastDigest 10x
- 6 µL H2O
- Mix for BBa_K1707011 and BBa_K1707012:
- 15 µL plasmid
- 1 µL XbaI
- 1 µL PstI
- 2 µL Buffer FastDigest 10x
- 1 µL H2O
- Mix for BBa_R0040 and BBa_I13602:
- 10 µL plasmid
- 1 µL EcoRI
- 1 µL XbaI
- 2 µL Buffer FastDigest 10x
- 6 µL H2O
- Mix for BBa_R0051
- 10 µL plasmid
- 1 µL SpeI
- 2 µL PstI
- 2 µL Tango Buffer 10x
- 6 µL H2O
Incubation 1h30, 37°C
- Mix for BBa_K1707004:
- 2 µL Tango Buffer 10x
- 1 µL SpeI
- 2 µL H2O
- 15 µL plasmid
Incubation 1h30, 37°C After incubation, we add:
- 3 µL Tango Buffer 10x
- 1 µL EcoRI
- 1 µL H2O
Incubation 1h30, 37°C
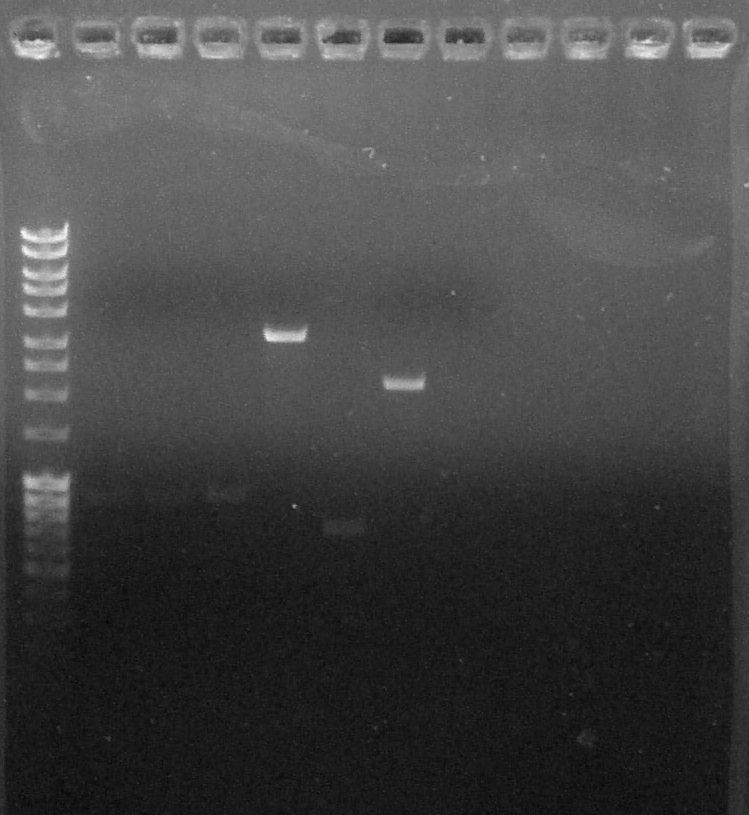
Purification by Electrophoresis
by Coralie
Agarose gel 1%, 100V
Biobricks:
- BBa_K1707004 #1 and #2 (digested by EcoRI + SpeI)
- BBa_K1707011 #1 (digested by XbaI +PstI)
- BBa_K1707006 #1 (digested by XbaI + PstI)
- BBa_K1707012 #1 (digested by XbaI + PstI)
- BBa_S03518 #1 (digested by XbaI + PstI)
Verification of gel purification, from left to right: 1. Empty, 2. DNA Ladder, 3. BBa_K1707004#1, 4. BBa_K1707004#2, 5. BBa_K1707006, 6. BBa_K1707011, 7. BBa_K1707012, 8. BBa_S03518, 9. Empty, 10. Empty
We can conclude that:
- BBa_K1707004 #1 and #2 are OK, we can cut the band
- BBa_K1707006 is OK, we can cut the band
- BBa_K1707011: we can't see anything on the gel
- BBa_K1707012: we can't see anything on the gel
- BBa_S03518: is OK, we can cut the band
Purification
by Pauline
Biobricks:
- BBa_K1707004 #2
- BBa_K1707011 #1
- BBa_K1707006 #1
- BBa_K1707012 #1
- BBa_S03518 #1
- BBa_R0040 #1
- BBa_I13602 #1
- BBa_R0051 #1
With Macherey-Nagel Purification kit
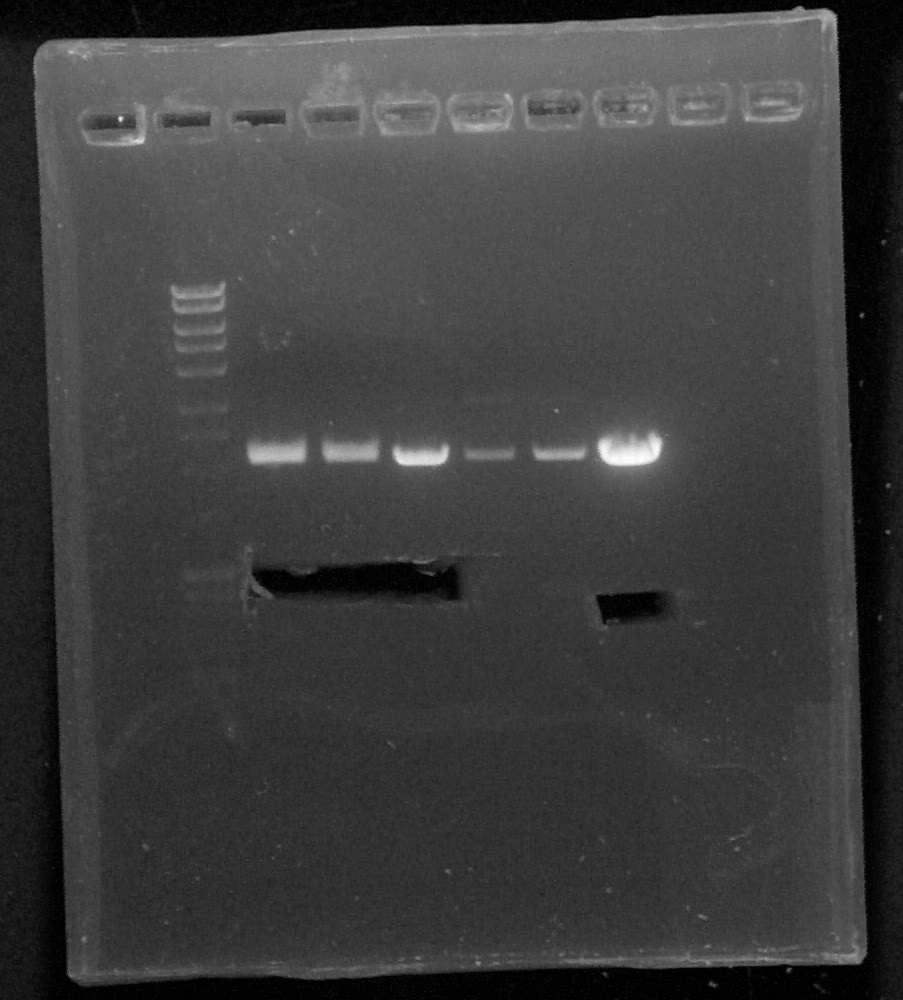
Quantification
by Pauline
Biobricks:
- BBa_K1707004 #2 x2
- BBa_K1707006 #1
- BBa_S03518 #1
- BBa_R0040 #1
- BBa_I13602 #1
- BBa_R0051 #1
Agarose gel 1%, migration at 100V
Quantification, from left to right: 1. DNA Ladder, 2. BBa_K1707004#1, 3. BBa_K1707004#2, 4. BBa_K1707006, 5. BBa_I13602, 6. BBa_S03518, 7. BBa_R0040, 8. BBa_R0051, 9. Empty, 10. Empty, 11. Empty, 12. Empty
We can conclude:
- BBa_K1707004 #2: 20µg/µL
- BBa_K1707006 #1: 30µg/µL
- BBa_S03518 #1: 50µg/µL
- BBa_R0040 #1: 30 µg/µL
- BBa_I13602 #1: 50µg/µL
- BBa_R0051 #1: we can't see anything
New Culture
by Pauline
Biobricks:
- BBa_K1707026
- BBa_K1707032
We put two clones of each in 5mL LB + 5µL Chloramphenicol
Member present:
- Instructors: Claire
- Students: Coralie and Pauline