Difference between revisions of "Team:Waterloo/Design"
PeterHoang (Talk | contribs) |
|||
| Line 10: | Line 10: | ||
[Lab paragraph] | [Lab paragraph] | ||
</p> | </p> | ||
| − | <p> | + | <p>The ability re-purpose sgRNAs to guide Cas9 to new target sites currently requires re-cloning the entire 500 base pair sgRNA sequence . By inserting restriction enzyme sites flanking the 20 base pair guide sequence of sgRNA, researchers will have the ability to more easily swap in new targets for Cas9. By taking advantage of the existence of a single "G" nucleotide at the bottom of the first stem-loop that doesn't follow complementarity with the opposing nucleotide in the sgRNA scaffold, we were able to insert a restriction enzyme site without also introducing that site downstream in the original DNA sequence. We then placed another restriction site after the U6 promoter but before the gRNA to complete the swap. Our experiments showed that this modified version of the sgRNA was still capable of working to silence gene expression with a dCas9 in similar level as the none modified.</p> |
| − | + | ||
| − | + | ||
| − | + | ||
<section id="cas9" title="Cas9 PAM Flexibility" class="cas9mod"> | <section id="cas9" title="Cas9 PAM Flexibility" class="cas9mod"> | ||
Revision as of 03:52, 19 September 2015
Design
Simple sgRNA Exchange
[Lab paragraph]
The ability re-purpose sgRNAs to guide Cas9 to new target sites currently requires re-cloning the entire 500 base pair sgRNA sequence . By inserting restriction enzyme sites flanking the 20 base pair guide sequence of sgRNA, researchers will have the ability to more easily swap in new targets for Cas9. By taking advantage of the existence of a single "G" nucleotide at the bottom of the first stem-loop that doesn't follow complementarity with the opposing nucleotide in the sgRNA scaffold, we were able to insert a restriction enzyme site without also introducing that site downstream in the original DNA sequence. We then placed another restriction site after the U6 promoter but before the gRNA to complete the swap. Our experiments showed that this modified version of the sgRNA was still capable of working to silence gene expression with a dCas9 in similar level as the none modified.
Cas9 PAM Flexibility
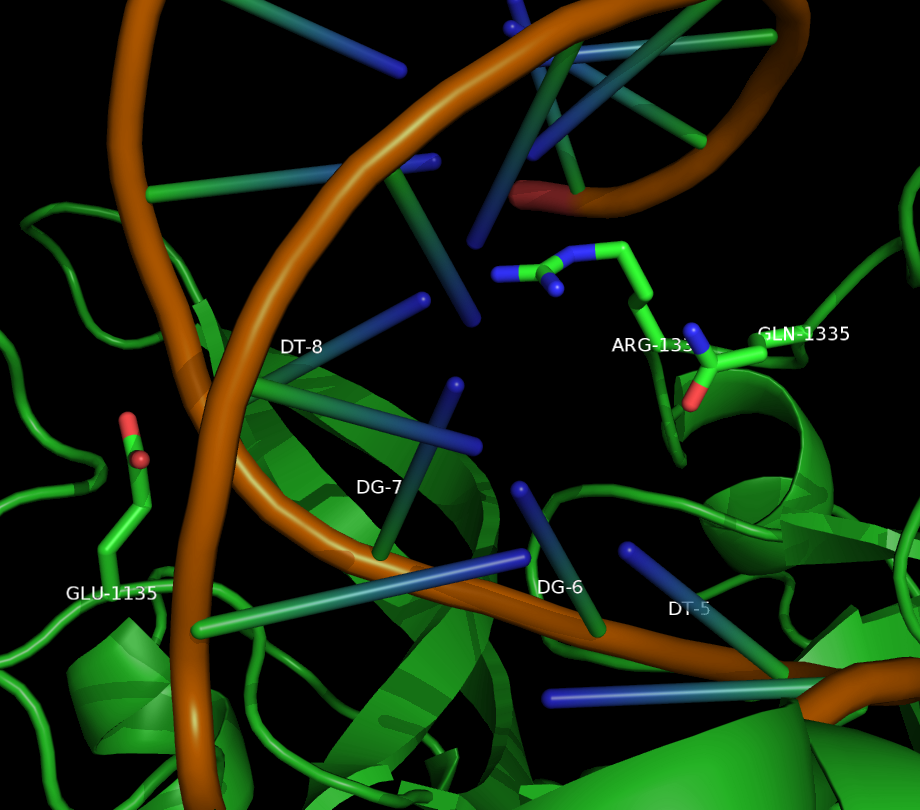
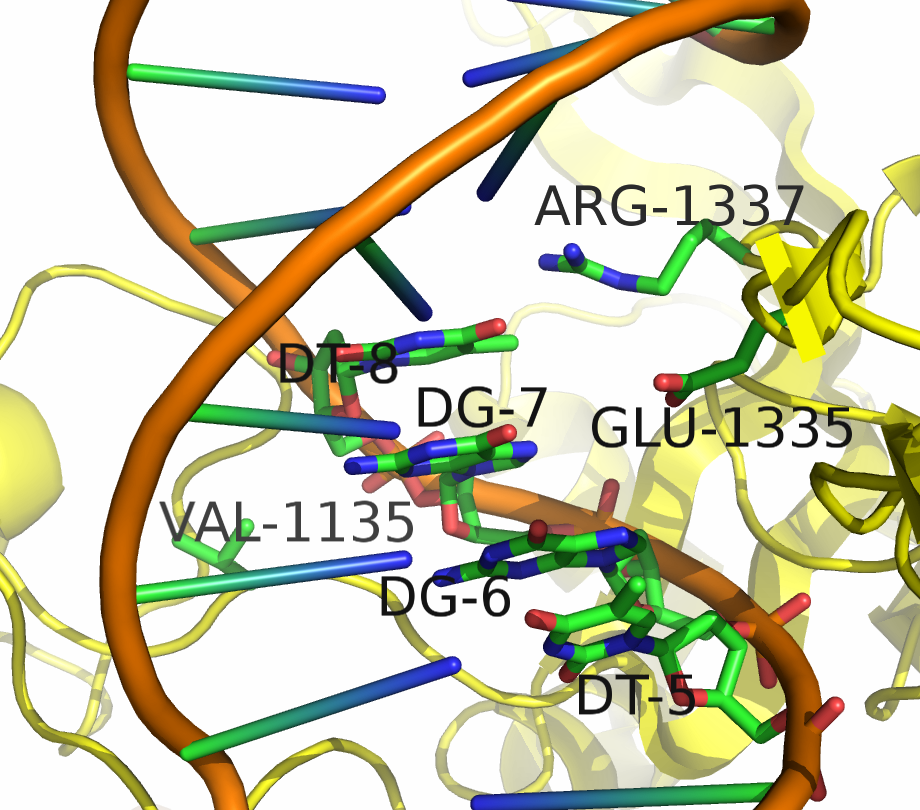
Being able to target DNA at different protospacer adjacent motif (PAM) site will allow scientists to target a larger portion of the genome. "Dead" Cas9 (dCas9) allows for easier analysis of products because the DNA still remains intact. Kleinstiver et al proposed an EQR variant of Cas9 with three amino acid substitutions. This new proposed version of Cas9 targeted an NGAG PAM sequence. Our design was to make these three amino acid substitutions in dCas9 and try to target an NGAG PAM site in GFP.
Kleinstiver et al. recently demonstrated modified spCas9 with altered PAM specificity . Their results motivated us to explore computational methods of assessing spCas9 mutants for altered PAM specificity profiles. We created an analysis pipeline using python that makes use of PyRosetta, a well known molecular dynamics toolkit. Our suite of scripts is described in our Modeling and Software pages.

CRISPR Plant Defense
[Lab paragraph]
[Math paragraph]
To model our antiviral application, we looked at the antiviral effects of CRISPR/Cas9 targeting on three scales: CaMV genomes, plant cells and plant leaves. Details of our modeling approach, and how it influenced our design, are provided in our Modeling pages.



References
By talking about your design work on this page, there is one medal criterion that you can attempt to meet, and one award that you can apply for. If your team is going for a gold medal by building a functional prototype, you should tell us what you did on this page. If you are going for the Applied Design award, you should also complete this page and tell us what you did.
Note
In order to be considered for the Best Applied Design award and/or the functional prototype gold medal criterion, you must fill out this page.
This is a prize for the team that has developed a synthetic biology product to solve a real world problem in the most elegant way. The students will have considered how well the product addresses the problem versus other potential solutions, how the product integrates or disrupts other products and processes, and how its lifecycle can more broadly impact our lives and environments in positive and negative ways.
If you are working on art and design as your main project, please join the art and design track. If you are integrating art and design into the core of your main project, please apply for the award by completing this page.