Difference between revisions of "Team:Nagahama/Design"
(Prototype team page) |
|||
| Line 1: | Line 1: | ||
{{Nagahama}} | {{Nagahama}} | ||
| + | |||
| + | |||
| + | |||
| + | = '''Protocols''' = | ||
| + | Our Lab's Protocols | ||
| + | |||
| + | == Medium == | ||
| + | |||
| + | === LB medium (100 mL liquid) === | ||
| + | |||
| + | 1.Measure 1g Tripton | ||
| + | |||
| + | 2.Measure 0.5g Yeast Extract | ||
| + | |||
| + | 3.Measure 1g Nacl | ||
| + | |||
| + | 4.Add 100mL H2O | ||
| + | |||
| + | 5.autoclave(121℃ 20min) | ||
| + | |||
| + | === 2×YT medium (100mL liquid)=== | ||
| + | |||
| + | 1.Measure 1.6g Tripton | ||
| + | |||
| + | 2.Measure 1g Yeast Extract | ||
| + | |||
| + | 3.Measure 0.5g Nacl | ||
| + | |||
| + | 4.Add 100mL H2O | ||
| + | |||
| + | 5.autoclave(121℃ 20min) | ||
| + | |||
| + | |||
| + | == DNA work == | ||
| + | |||
| + | |||
| + | === Agarose gel(100mL) === | ||
| + | |||
| + | Method of Making 0.7% Agarose gel | ||
| + | |||
| + | 1.Measure 0.7g Agarose | ||
| + | |||
| + | 2.Add 100mL TAE buffer | ||
| + | |||
| + | 3.Heat(till agarose melted)*We used a microwave oven. | ||
| + | |||
| + | 4.Pur agarose into a gel maker | ||
| + | |||
| + | 5.Set a comb | ||
| + | |||
| + | 6.Wait till agarose curdles | ||
| + | |||
| + | 7.Pull an comb | ||
| + | |||
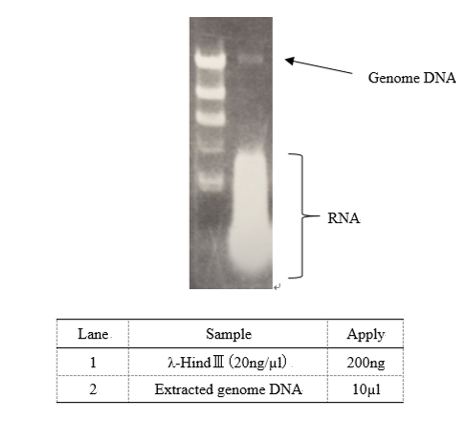
| + | === Genome DNA extraction === | ||
| + | ↓Cultivate ''E. coli DH5α'' using LB medium 2ml×2 tubes O/N | ||
| + | |||
| + | ↓Centrifuge culture 1.5ml (13,000rpm 4℃ 1min) | ||
| + | |||
| + | ↓Remove the culture | ||
| + | |||
| + | ↓+TNE buffer 1.0ml (10mM Tris pH 8.0, 10 mM NaCl, 10 mM EDTA) | ||
| + | |||
| + | ↓vortex | ||
| + | |||
| + | ↓Centrifuge cell suspension (13,000rpm 4℃ 2min) | ||
| + | |||
| + | ↓Remove supernatant | ||
| + | |||
| + | ↓+TNE buffer including 1% Triton X-100 270µl | ||
| + | |||
| + | ↓Suspend cell gently | ||
| + | |||
| + | ↓+5mg/ml Lysozyme solution 30µl (0.15g Lyzozyme + 30µl sterile water) | ||
| + | |||
| + | ↓Reaction 37℃ 30min | ||
| + | |||
| + | ↓+ 20mg/ml Proteinase K (fin.con: 1mg/ml) TaKaRa Code:9033 | ||
| + | |||
| + | ↓Reaction 65℃ 2h | ||
| + | |||
| + | ↓+Phenol chloroform 300µl ...(1) | ||
| + | |||
| + | ↓Mix the solution gently ...(2) | ||
| + | |||
| + | ↓Centrifuge (13,000rpm 4℃ 8min) ...(3) | ||
| + | |||
| + | ↓Transfer only water layer to new 1.5ml tube ...(4) | ||
| + | |||
| + | ↓Repeat (1)~(4) 2 times | ||
| + | |||
| + | ↓+3M Sodium acetate 30µl | ||
| + | |||
| + | ↓Mix gently | ||
| + | |||
| + | ↓+99.5% EtOH 750µl | ||
| + | |||
| + | ↓Mix gently | ||
| + | |||
| + | ↓Centrifuge (13,000rpm 4℃ 10min) | ||
| + | |||
| + | ↓Remove supernatant | ||
| + | |||
| + | ↓+70% EtOH 500µl | ||
| + | |||
| + | ↓Centrifuge (13,000rpm 4℃ 1min) | ||
| + | |||
| + | ↓Dry up the pellet covering with aluminum foil at room temperature 30min | ||
| + | |||
| + | ↓+TE buffer 50µl | ||
| + | |||
| + | ↓Suspend DNA gently | ||
| + | |||
| + | [[File:GenomeDNA_DH5a2.png]] | ||
| + | |||
| + | === Plasmids extraction === | ||
| + | |||
| + | |||
| + | |||
| + | === PCR === | ||
| + | |||
| + | === Transformation by heat shock=== | ||
| + | |||
| + | == isoprenoid production == | ||
| + | |||
| + | == geraniol tolerant assay == | ||
| + | |||
| + | == GC == | ||
| + | |||
| + | == GC-MS == | ||
| + | |||
| + | |||
<html> | <html> | ||
| − | <h2> | + | <h2>Experiments & Protocols</h2> |
| − | <p> | + | <p>Describe the experiments, research and protocols you used in your iGEM project.</p> |
| − | + | ||
| − | </p> | + | |
| − | < | + | <h5>What should this page contain?</h5> |
| − | < | + | <ul> |
| − | < | + | <li> Protocols </li> |
| − | </ | + | <li> Experiments </li> |
| + | <li>Documentation of the development of your project </li> | ||
| + | </ul> | ||
| − | |||
| − | |||
| − | |||
| − | |||
| + | <h4>Inspiration</h4> | ||
| + | <ul> | ||
| + | <li><a href="https://2014.igem.org/Team:Colombia/Protocols">2014 Colombia </a></li> | ||
| + | <li><a href="https://2014.igem.org/Team:Imperial/Protocols">2014 Imperial </a></li> | ||
| + | <li><a href="https://2014.igem.org/Team:Caltech/Project/Experiments">2014 Caltech </a></li> | ||
| + | </ul> | ||
</div> | </div> | ||
| − | |||
</html> | </html> | ||
Revision as of 05:39, 7 September 2015
Contents
Protocols
Our Lab's Protocols
Medium
LB medium (100 mL liquid)
1.Measure 1g Tripton
2.Measure 0.5g Yeast Extract
3.Measure 1g Nacl
4.Add 100mL H2O
5.autoclave(121℃ 20min)
2×YT medium (100mL liquid)
1.Measure 1.6g Tripton
2.Measure 1g Yeast Extract
3.Measure 0.5g Nacl
4.Add 100mL H2O
5.autoclave(121℃ 20min)
DNA work
Agarose gel(100mL)
Method of Making 0.7% Agarose gel
1.Measure 0.7g Agarose
2.Add 100mL TAE buffer
3.Heat(till agarose melted)*We used a microwave oven.
4.Pur agarose into a gel maker
5.Set a comb
6.Wait till agarose curdles
7.Pull an comb
Genome DNA extraction
↓Cultivate E. coli DH5α using LB medium 2ml×2 tubes O/N
↓Centrifuge culture 1.5ml (13,000rpm 4℃ 1min)
↓Remove the culture
↓+TNE buffer 1.0ml (10mM Tris pH 8.0, 10 mM NaCl, 10 mM EDTA)
↓vortex
↓Centrifuge cell suspension (13,000rpm 4℃ 2min)
↓Remove supernatant
↓+TNE buffer including 1% Triton X-100 270µl
↓Suspend cell gently
↓+5mg/ml Lysozyme solution 30µl (0.15g Lyzozyme + 30µl sterile water)
↓Reaction 37℃ 30min
↓+ 20mg/ml Proteinase K (fin.con: 1mg/ml) TaKaRa Code:9033
↓Reaction 65℃ 2h
↓+Phenol chloroform 300µl ...(1)
↓Mix the solution gently ...(2)
↓Centrifuge (13,000rpm 4℃ 8min) ...(3)
↓Transfer only water layer to new 1.5ml tube ...(4)
↓Repeat (1)~(4) 2 times
↓+3M Sodium acetate 30µl
↓Mix gently
↓+99.5% EtOH 750µl
↓Mix gently
↓Centrifuge (13,000rpm 4℃ 10min)
↓Remove supernatant
↓+70% EtOH 500µl
↓Centrifuge (13,000rpm 4℃ 1min)
↓Dry up the pellet covering with aluminum foil at room temperature 30min
↓+TE buffer 50µl
↓Suspend DNA gently
Plasmids extraction
PCR
Transformation by heat shock
isoprenoid production
geraniol tolerant assay
GC
GC-MS
Experiments & Protocols
Describe the experiments, research and protocols you used in your iGEM project.
What should this page contain?
- Protocols
- Experiments
- Documentation of the development of your project