Difference between revisions of "Team:Czech Republic/Microfluidics"
(→References) |
(→Soft-lithography) |
||
| Line 32: | Line 32: | ||
[[File:Microfluidic_chips.jpg|thumbnail|Fabricated microfluidic devices (three PDMS molds bonded to single glass slide)]] | [[File:Microfluidic_chips.jpg|thumbnail|Fabricated microfluidic devices (three PDMS molds bonded to single glass slide)]] | ||
| − | Microfluidic channels were formed using PDMS soft-lithography technology, which has proven biocompatibility and can be readily applied in available laminar flow cabinets. Photomask and silicon master fabrication was outsourced. Fabrication of microfluidic devices was divided in two subsequent steps. In the first step, silicon masters were used for the PDMS molding. In the second step, PDMS molds were bonded to the glass substrates to form encapsulated microfluidic devices using air plasma technology. | + | Microfluidic channels were formed using PDMS soft-lithography technology, which has proven biocompatibility and can be readily applied in available laminar flow cabinets {{:Team:Czech_Republic/Template:ReferenceRef|Fikar2015}}. Photomask and silicon master fabrication was outsourced. Fabrication of microfluidic devices was divided in two subsequent steps. In the first step, silicon masters were used for the PDMS molding. In the second step, PDMS molds were bonded to the glass substrates to form encapsulated microfluidic devices using air plasma technology. |
=== PDMS molding === | === PDMS molding === | ||
Revision as of 12:14, 15 September 2015
Microfluidics
Contents
Abstract
Microfluidic devices were designed and fabricated to characterise the developed IOD band system. Signal transduction between transmitting and receiving cells was characterised using spatially separated cell cultures inside single microfluidic channel in conjunction with live fluorescence microscopy. In addition, agglutination tests were performed on-chip to decrease the time necessary in standard agglutination tests and enable dynamic control of the agglutination process.
Key Achievements
- Set of microfluidic devices fabricated by PDMS soft-lithography.
- Signal transduction dynamically tested between several types of cell in a microfluidic system.
- Demonstrated on-chip induced yeast agglutination on-chip achieved by complementary EpCam and Anti-EpCam molecules displayed on cell surface by Yeast Surface Display.
- Demonstrated yeast induced blood agglutination on-chip by human antigen A production.
Introduction
The diffusion processes are slow, and the inertial effects are negligible on micro-scale with low Reynolds number [Angelescu2011] [Nam2002]. Hence microfluidics enables complex control of the cellular microenvironment. Microfluidic experiments in conjunction with live fluorescence microscopy were designed and performed to verify and characterise the developed IOD system, the signal transduction, and the agglutination mechanism.
Soft-lithography
Microfluidic channels were formed using PDMS soft-lithography technology, which has proven biocompatibility and can be readily applied in available laminar flow cabinets [Fikar2015]. Photomask and silicon master fabrication was outsourced. Fabrication of microfluidic devices was divided in two subsequent steps. In the first step, silicon masters were used for the PDMS molding. In the second step, PDMS molds were bonded to the glass substrates to form encapsulated microfluidic devices using air plasma technology.
PDMS molding
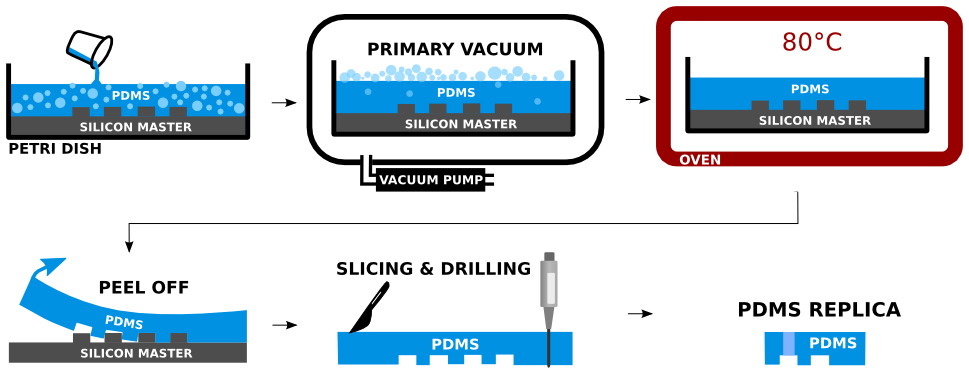
Two part silicone elastomer Sylgard 184 is used to produce PDMS. The base part is mixed with sufficient amount of curing agent (10:1 ratio). The mixture is centrifuged to remove the air bubbles introduced by the mixing. The Si master is placed in an aluminum foil container and the mixture is poured over. The remaining air bubbles are removed from the PDMS by sharp tip of a needle. The poured PDMS is maintained in perfect horizontal position to assure good planarity, and is cured in an oven, for 2 hours at 80°C. The PDMS edges are cut off with sharp tool and the PDMS is peeled off the Si master. The PDMS mold is sliced into sections containing individual devices. Inlets and outlets are drilled carefully by biopsy punch of the appropriate diameter at the desired locations of the PDMS replica. Detailed experimental protocol is provided here.
Bonding of PDMS to the glass substrate
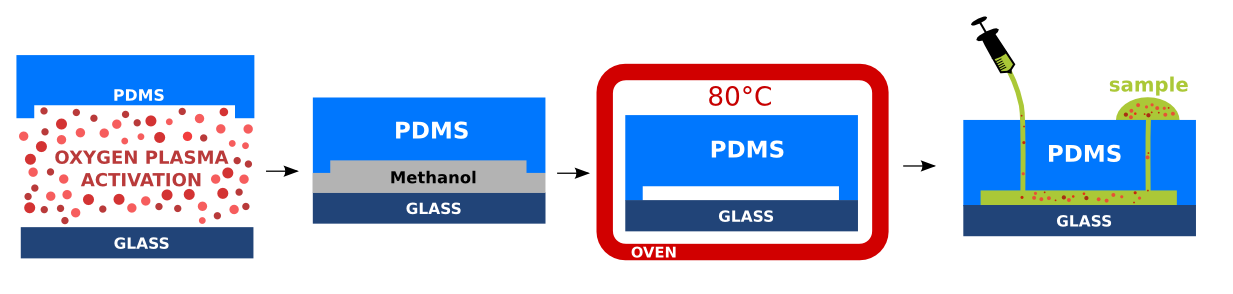
Prepared PDMS replicas with imprinted micro-structures are cleaned properly with scotch tape. The PDMS and glass substrate are treated by air plasma for 2.5 minutes. The air plasma affects the PDMS backbone and forms reactive silanol functional groups (Si-OH) enabling formation of permanent irreversible covalent bond of the PDMS to the glass substrate [Wong2009]. In addition, the PDMS treatment with air plasma is beneficial as it avoids nonspecific adsorption, decreases cell clogging and turns the PDMS to hydrophilic. The hydrophilicity of PDMS facilitates the future microchannel wetting [Kalio2006]. Immediately after the air plasma treatment, the glass substrate is brought into contact with the PDMS replica and placed in the oven, for 5 minutes at 80°C. The bonded devices were stored in a room temperature. Inlets and outlets were sealed with scotch tape to avoid contamination. Detailed experimental protocol is provided here.
Experimental setup
Microfluidic experiments were conducted on a platform already established by the Georgiev lab. The laboratory is equipped with precise microfluidic syringe system (neMESYS Low Pressure Syringe Pumps), and microscopic station enabling fluorescence imaging and live cell microscopy (Olympus IX83).
Microfluidic experiments
Signal transduction distance and dynamics
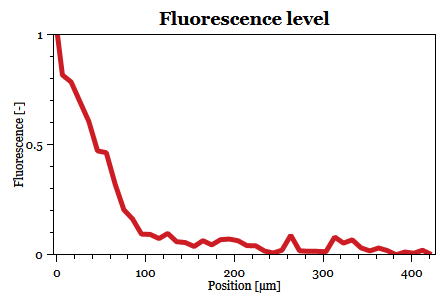
Fabricated microfluidic devices were used to characterise the signal transduction between two types of cells. The first type of cell transmits signal in the form of pheromone to the receiving cell type. Both cell types are introduced to the microfluidic device in parallel through separated inlets. The receiving cell contains synthetic reporter plasmid responsible for expression of Green Fluorescent Protein (GFP) when pheromone molecule reaches certain threshold and activates the MAPK cascade. Thanks to the laminar nature of the flow, the individual cell types remain spatially separated inside the microfluidic device. The spatial separation is well visible during the filling of the microfluidic device. The flow is stopped when the filling of the microfluidic device is finished, and the rest of the experiment is performed statically. Time lapse fluorescence microscopy is used to dynamically measure levels of GFP expression in the cells that receive the signal. Results of the experiment show...
Agglutination on-chip
Standard agglutination tests are time consuming and require specialized equipment occupying large bench space [Choi2009]. Special microfluidic topologies were designed to test yeast induced agglutination on-chip to lower the time and sample volume requirements. Agglutination tests were performed with synthetic yeast strains inducing agglutination through epithelial-cell adhesion molecule (EpCAM) bonding to its antibody. Both of the molecules are displayed on yeast cell surface when induced and subsequently cause cell clumping which serves as easily observable visual feedback providing information about the IOD band test system.
Personnel
- Martin Cienciala - Microfluidic experiments
- Vaclav Pelisek - Fabrication of microfluidic devices
- Pavel Fikar - Scientific advisor
References
- ↑ Dan E. Angelescu (2011). Highly Integrated Microfluidic Design. Artech House, Norwood.
- ↑ Nam-Trung Nguyen and Steven T Wereley (2002). Fundamentals and applications of microfluidics. Artech House.
- ↑ Ieong Wong (2009). Surface molecular property modifications for poly(dimethylsiloxane) (pdms) based microfluidic devices. Microfluid Nanofluid, 7:291–306.
- ↑ Johana Kuncova-Kallio (2006). Pdms and its suitability for analytical microfluidic devices. In Proceedings of the 28th IEEE EMBS Annual International Conference, New York City, USA, 9, IEEE.
- ↑ Sung Hwan Choi (2009). Microinjection molded disposable microfluidic lab-on-a-chip for efficient detection of agglutination. Microsystem Technologies, 15(2):309-316.