Difference between revisions of "Team:Nagahama/Experiments"
(→Why Flavorator?) |
(→Why Flavorator?) |
||
| Line 34: | Line 34: | ||
'''Fragrance of wasabi''' | '''Fragrance of wasabi''' | ||
| + | |||
| + | Rice cake | ||
[[File:もち.jpg|サイズpx|thumb|center|図の説明]] | [[File:もち.jpg|サイズpx|thumb|center|図の説明]] | ||
Revision as of 10:47, 13 September 2015
Contents
Result and Discussion
POC
Why Flavorator?
30 years ago, “KOZOKO” was proposed by Professor Yozo Iwanami “KOZOKO” is a box to preserve food in a fragrance. Then do you think what fragrance we does fill? It’s plant’s one. Such fragrance has antibacterial and/or bacteriostatic activities. Thus, “KOZOKO” would become an energy-saving substitute for an electric refrigerator. Although, it has two problem to realize “KOZOKO”. One, there is a lot of plants to produce fragrance. The other, it take high cost to extraction of fragrance component from plants. Resulting, it was not popularly used.
Today, we create modern version “KOZOKO” by synthetic biological technique. We named it “Flavorator” Recombinant E. coli produces antimicrobial and/or bacteriostatic volatile gaseous substances that suppress bacterial unwanted growth in the “Flavorator”. Our choice was geraniol and farnesol. Geraniol and farnesol are the major constituents of rose fragrance. These fragrances not only emit fragrant odor to humans but also kill/or suppress bacterial cells as well as insects such as fruit fly
Then we designed three points in order to create “Flavorator”.
Point 1 is A lot of terpenoid's precursor
Point 2 is Produce geraniol and farnesol
Point 3 is Release the products and Increase the antibacterial resistance
In this way, we are sure that these functions can avoid food decay and solve the food problem.
We examined our working hypothesis to “Flavorator” that the volatile gaseous substances from plants’ origin can show either the antibacterial or bacteriostatic activity in a box like Kozoko.
Fragrance of Garlic
We prepared 20g pork and 5g garlic. We left the pork for an hour on the table and grated garlics. We spread the garlic grated on the pork (A). We left the two pork in the box at 18℃ for 2 month. Left pork (A) didn’t change the color. Right pork (B) changed Pink to Brown. We found from this result that fragrance of plants have antibacteril volatiles.
Fragrance of wasabi
Rice cake
Fragrance of Rose

先駆体合成 (キノン)
GOH生産
FOH生産
ispA+MEP.dev[http://parts.igem.org/wiki/index.php?title=Part:BBa_K1653025]
λPL+r.b.s.+ispA+MEP+×× (r.b.s.+dxs+r.b.s.+m-idi+r.b.s.+ispDF)
E. coli strain engineered with MEP pathway enzymes, ispDF, idi, and dxs , in combination with the enzyme gens, ispA, produced farnesol (Fig. 4A), which was detected by the Gas chromatography/Mas (Fig. 4B), having the same retention time as the farnesol chemica sample (Fig. 4A), while the counterpart control E. coli did not produce farnesol under the same conditions (Fig. 4C). Neither E. coli engineered with MEP pathway enzymes only nor the one engineered ispA only showed any farnesol by the Gas chromatography/Mass (Figs. 4D and E). Farnesol is generated through hydrolysis of farnesyl diphosphate (FPP) by the endogenous phosphatases. Increase in farnesol should be associated with an increased intracellular FPP level. FPP is, in turn, converted from geranyl diphosphate (GPP), whose precursors are IPP and DMAPP. IPP and DMPP are end products of MEP pathway that exists in E. coli. Conversion to FPP from IPP or DMPP requires ispA (or m-ispA). Following this context, we speculate that E. coli could produce farnesol better than the counterpart control cells under the up-regulated cellular conditions of an increased intracellular MEP pathway enzymes by metabolic engineering in combination with the special enzyme that converts IPP or DMAPP into FPP.
Gas Chromatography/Mass(GC/MS)
Fig4:The FOH standard solution (Ref) was used as a control. The peak corresponding to the FOH standard at 8.5 min is indicated by an arrow. The peak at 8.5 min was applied to GC/MS. The FOH standard solution (Ref) was used as a control. E. coli JM109(Bba_K165025) were compared with respect to FOH formation using GC-MS.
排出
耐性
Production of Geraniol and Farnesol in E. coli
GC
GC-MS
New Part2
ispA+MEP.dev[http://parts.igem.org/wiki/index.php?title=Part:BBa_K1653025]
λPL+r.b.s.+ispA+MEP+×× (r.b.s.+dxs+r.b.s.+m-idi+r.b.s.+ispDF)
FOH is probably generated through FPP hydrolysis by endogenous phosphatases, which are induced by an increased intracellular FPP level Analogously, we hypothesized that E. coli could produce FOH under cellular conditions of an increased intracellular FPP level through metabolic engineering. A MEP pathway has been shown to synthesize IPP and DMAPP efficiently in E. coli. Because of its high hydrophobicity and low volatility, decane was chosen to extract and solubilize FOH from culture broth. The decane overlay in the two-phase culture did not affect growth, and FOH could be solubilized in the decane phase with negligible volatile loss. We adopt 1 mL of decane overlaid to 5 mL of culture broth. Two-phase culture of E. coli JM109 (BBa_K165025) was carried out in 2YT medium containing 1% glycerol at 29°C for 48 h. The decane phase of the two-phase culture was collected to analyze the FOH content by GC-MS. In the GC-MS analysis (Fig. 4A-G), there was a main peak at 8.5 min in the collected decane phase sample, which corresponded to the reference solution of the standard FOH compound dissolved in decane. Mass spectrometry confirmed that the peak at 8.5 min was FOH (Fig. 4-A). However, the peak was not observed in two-phase culture without introducing BBa_K165025. The formation of FOH from FPP was further confirmed by blocking FPP synthesis. In the GC-MS, the FOH peak was observed in E. coli JM109 (BBa_K165025) culture, whereas no peak was observed with transformed E. coli JM109. It was found that FOH need not only ispA(BBa_K165018) but also MEP(BBa_K165024) in E. coli.We submit new part(BBa_K165025) as producing FOH.
Gas Chromatography/Mass(GC/MS)
Fig4:The FOH standard solution (Ref) was used as a control. The peak corresponding to the FOH standard at 8.5 min is indicated by an arrow. The peak at 8.5 min was applied to GC/MS. The FOH standard solution (Ref) was used as a control. E. coli JM109(Bba_K165025) were compared with respect to FOH formation using GC-MS.
Geraniol transport assay
Transporting geraniol device
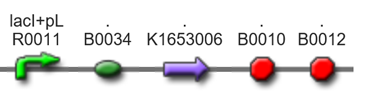 [http://parts.igem.org/wiki/index.php?title=Part:BBa_K1653020 Part's Page]
[http://parts.igem.org/wiki/index.php?title=Part:BBa_K1653020 Part's Page]

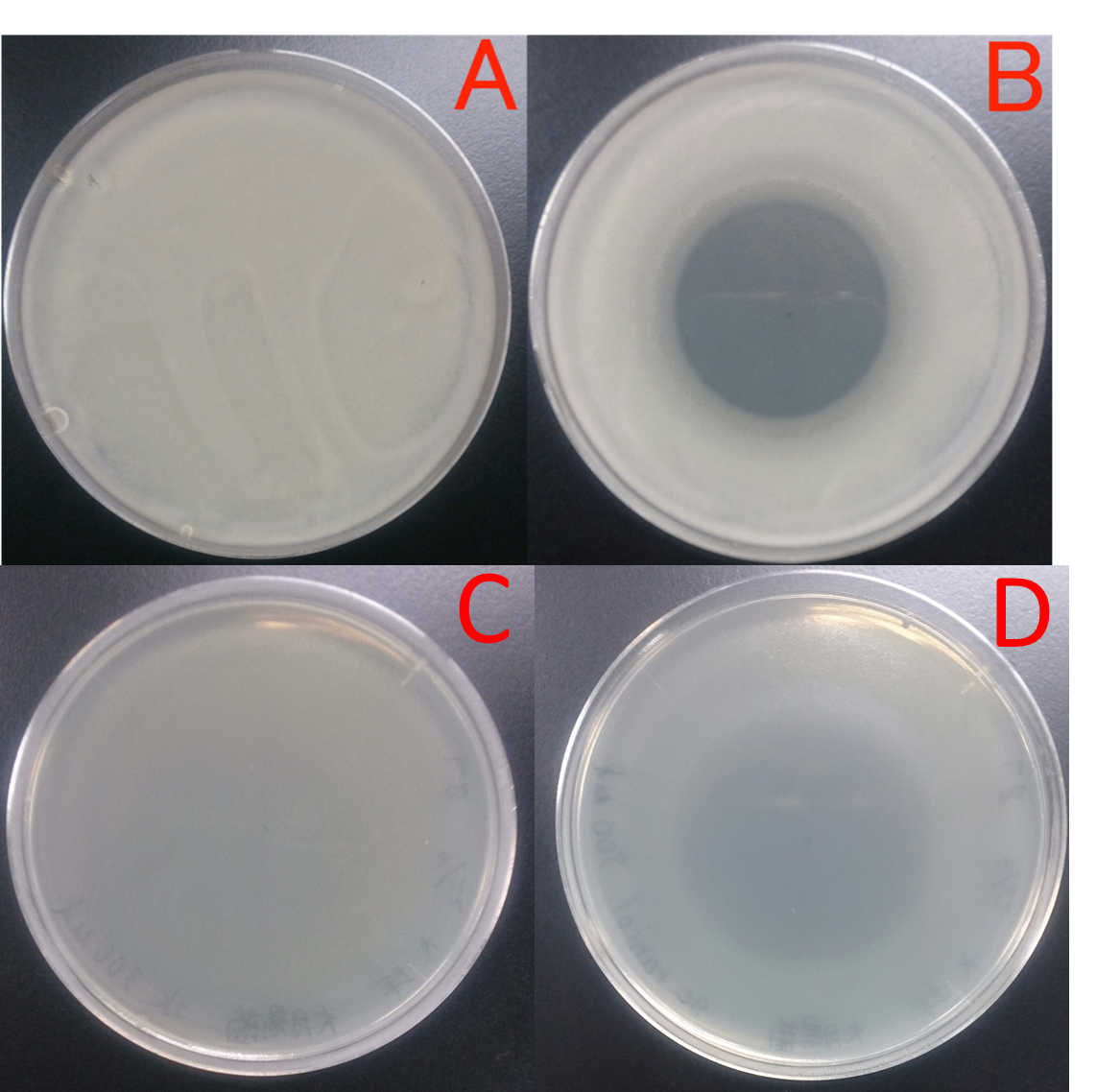
E. coli JM109 and E. coli JM109 (marA) were spotted on LBGMg agar plates in serial ten-fold dilutions (10‐¹~10⁻⁵), overlaid with geraniol solutions, and incubated at 30°C for 24 h. This figure shows that E. coli JM109 (marA) that overxpresses marA is more surviving on geraniol overlay plates than E. coli JM109 wild type.

A: E. coli JM109 (WT) + hexane
B: E. coli JM109 (marA) +hexane
C: E. coli JM109 (WT) + geraniol
D: E. coli JM109 (marA) +geraniol
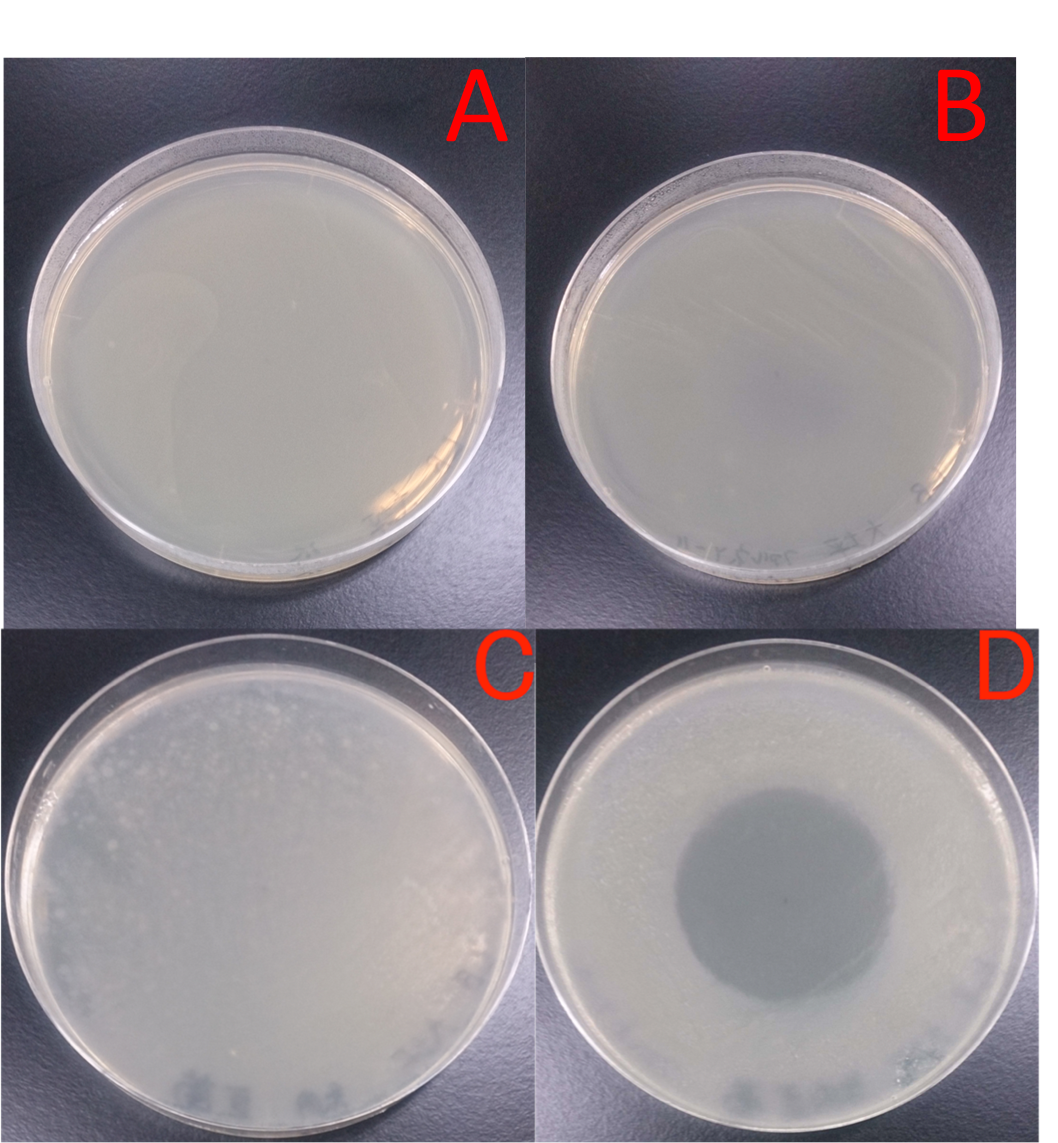
A and B increased almost the same on the plate. But C and D differed clealy. After 1 hour, C lost colony on the plate. In contrast, D could see colony all plates. This figure shows that E. coli JM109 (marA) that overxpresses marA is more surviving than E. coli JM109 wild type in the LBGMg midium overlayed geraniol solution.
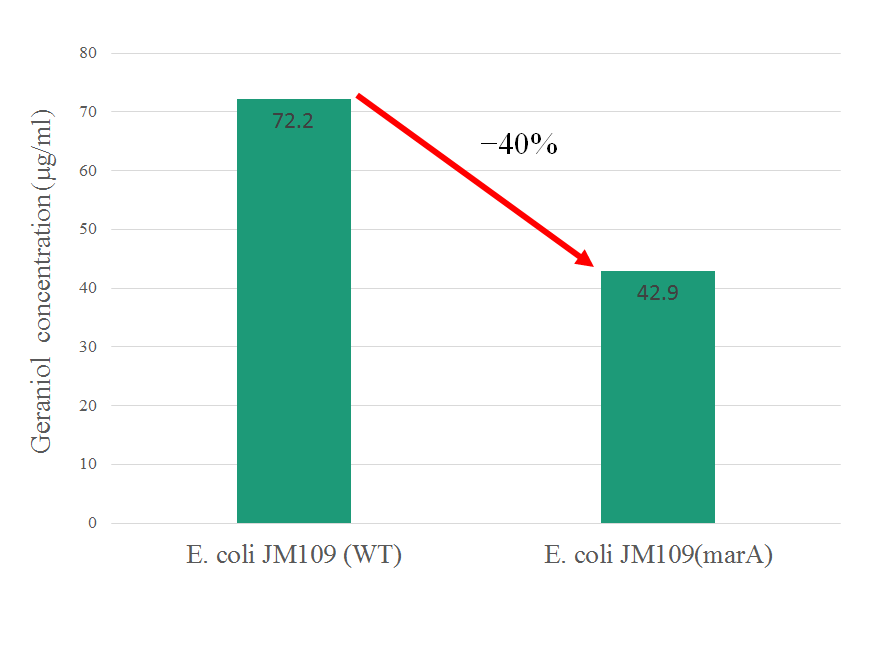
The intracellular geraniol concentration of E. coli JM109(marA) was observed at 42.9 μg/ml, which was 40% lower than 72.2 μg/ml of E. coli JM109 (WT). We confilmed that geraniol was transported out of the cell body by overexpressing of marA activates AcrAB-TolC multidrug efflux pump.
This is a new information about exsisting marA gene.
Gold Medal's requirement