Team:ETH Zurich/Project Description
- Project
- Modeling
- Lab
- Human
Practices - Parts
- About Us
A Microbial Beacon for Cancer Detection
Cancer as a major health concern
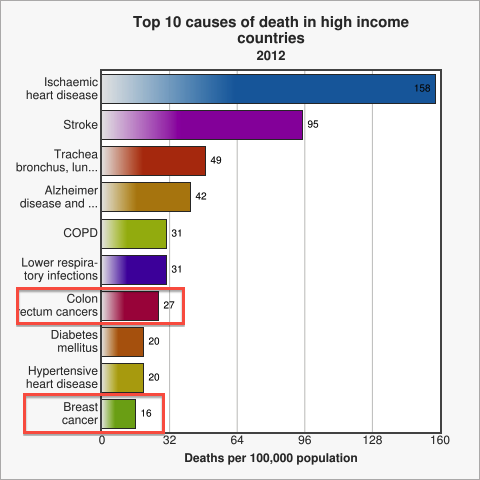
Cancer ranges amongst the top ten causes of death in developped countries [WHO]. Amongst all cancer patients, for approximately 90% the ultimate cause of death is metastasis [Saxe, 2013], be it during the initial occurence of cancer or at a time after successful treatment. Therefore, early diagnosis of metastasis is one of modern medicine's major and challenging problems. [WHO 2014] Thus, designing a novel method to test for circulating cancer cells, which represent the first sign of possible metastasis [Sleijfer 2007], quickly caught our attention as a potential project idea. After researching current testing methods we decided to come up with a system for the assessment of metastasis risk in cancer patients and high-risk groups using a genetically engineered strain of Escherichia coli. The big advanteage of a bacterial system over conventional analysis methods is the great capacity of signal integration in genetic circuits and the generation of a robust, yet sensitive output. Our engineered E. coli, called MicroBeacons, will provide a boradly applicable, cheap, and reliable alternative to existing test for circulating tumor cells (CTC).
A universally applicable system for CTC detection
Circulating tumour cells (CTCs) can be used as an indicator of the initial stages of metastasis.[Sleijfer 2007] In our project we aim to develop a novel method that is fast, economical, and reliable for the identification of CTCs by using MicroBeacons to detect two general cancer markers and indicate their cooccurence through fluorescence. By design, this makes our system applicable to many types of cancer. To facilitate handling the cells, our detection system will be embedded in a microfluidic chip built especially for this purpose. In contrast to existing methods which rely on detection of very specific cancer markers, like epithelial cell adhesion molecules [Cooke 2001, Garcea 2005, Singhal 2005] a single general test would be more economical if it can be mass produced and deployed in a cost-effective manner.
Using cancer cells' metabolic adaptation to our advantage
The first marker we are planning to detect is the increased production rate of lactate by cancer cells due to the Warburg effect, a metabolic shift occurring in tumor cells that is characterized by a much higher glycolysis rate and subsequent lactic acid production[Chen 2007, Vander 2009]. The increased output of L-lactate by cancer cells, that results from this shift, can be used to discriminate cancer cells from healthy cells. We found the lldPRD operon, which is endogenously present in E. coli to help it metabolize lactate, and decided to use this system to our advantage. The constitutive expression of LldR (BBa_K1847001), a repressor of the lldPRD operon in E. coli, is an important part of our system, since according to the literature, lldR acts as a repressor of promoters containing the respective lldR binding sites, the lldR operators O1 and O2. Upon exposure of E. coli to lactate, binding of lactate to LldR causes a conformational change, inhibiting lldR's ability to bind to its target operators, which in turn leads to activation of the operon[Aguilera 2008].
The concentration of lactate around the cells accumulates over time. Therefore, we designed our system to detect a fold-change using an incoherent feed-forward loop [Goentoro 2009]. Overall, an lldRO-lacO fusion promoter, which we designed specifically for this setup, acts as a filter, detecting our first general cancer marker and converting it into a signal that will be crucial for the next step of our design, which will thus only induce a final output if elevated lactate production rates are detected. This is where the second cancer marker comes into play.
Bacteria as apoptosis detectors
To make our system reliable, we neededa second general cancer marker which would form an AND gate with the first marker. We encountered the TNF-related apoptosis-inducing ligand (TRAIL), or sTRAIL in its soluble form, which induces apoptosis specifically in cancer cells, leaving healthy cells unharmed [Srivastava 2001]. The initiation of apoptosis can be detected by the strong binding (Kd=0.5-1 nM) of Annexin V to phosphatidylserine (PS) that appears on the outer membrane of the target CTCs in early stages of apoptosis[Engeland 1998]. Although Annexin V is an intracellular protein in mammalian cells[Hawkins 2002], we planned to engineer it to be expressed on the surface of our MicroBeacons, allowing them to bind to the target CTCs. Binding of several MicroBeacons on the surface of an apoptotic CTC will be detected via quorum sensing signals.

Figure 3. Annexin V crystallographic structure. Image from PDB
Higher selectivity thanks to AND gate
The elevated lactate production and binding of MicroBeacons on an apoptotic cell lead to a fluorescent signal. Both signals are chained in a way that quorum sensing can only be initiated upon detection of elevated lactate production rates. The final output of the system is the expression of reporter green fluorescent protein (GFP) coupled to this quorum sensing module, which is only be expressed in signifficant levels near a target if it is a cancer cell.
Overall, the integration of two general markers allows for the detection of various types of cancers with high reliability and a low false positive rate, while control of signal expression by the bacteria should result in a considerable increase in the signal to noise ratio compared to conventional assay kits. The direct binding of our MicroBeacons to cancer cells allows for the detection of lactate and AHL in a controlled microenvironment with minimal crosstalk from other cells in the sample or unbound MicroBeacons. Finally, the use of a fluorescent signal as our output signal will allow for easy measurement from the microfluidic chip through microscopy.
Glossary
Circulating Tumor Cell (CTC)
CTC are thought to be originated in the primary tumor from which they escape and colonize new tissues. They are believed to be the cause of metastasis. Therefore, they can be used as a marker for metastatic cancer [Cáceres, 2015].