Difference between revisions of "Team:Czech Republic/Microfluidics"
(→Key Achievements) |
(→Introduction) |
||
| Line 15: | Line 15: | ||
= Introduction = | = Introduction = | ||
| − | The diffusion processes are slow, and the inertial effects are negligible on micro-scale with low Reynolds number {{:Team:Czech_Republic/Template:ReferenceRef|Angelescu2011}}. Hence microfluidics enables complex control of the intercellular microenvironment. Microfluidic experiments in conjunction with live fluorescence microscopy were designed and performed to verify and characterize the signal transmission mechanism in the developed IOD band system. | + | The diffusion processes are slow, and the inertial effects are negligible on micro-scale with low Reynolds number {{:Team:Czech_Republic/Template:ReferenceRef|Angelescu2011}}. Hence microfluidics enables complex control of the intercellular microenvironment. Microfluidic experiments in conjunction with live fluorescence microscopy were designed and performed to verify and characterize the signal transmission mechanism in the developed IOD band system, and to verify the developed yeast-induced blood agglutination mechanism. |
= Soft-lithography = | = Soft-lithography = | ||
Revision as of 23:42, 18 September 2015
Microfluidics
Contents
Abstract
Microfluidic devices were designed and fabricated to characterize the signal transmission in the developed IOD band system. Signal transmission between transmitting and receiving cells was characterized using spatially separated cell cultures inside a single microfluidic channel in conjunction with live fluorescence microscopy. Plasmids coding for a synthetic reporter protein were transformed into signal-receiving cells to show activation of its yeast pheromone pathway. The on-chip characterization was used to evaluate the signal transmission range and its evolution in time. In addition, special microfluidic topologies were designed and fabricated to test yeast-induced agglutination on-chip to lower the time and sample volume requirements. Agglutination tests were performed with synthetic yeast strains inducing blood agglutination by displaying the human antigen A antibody (anti-HuA) on its surface. The yeast strains were designed and synthesised by Module 3. The amount of blood necessary for the experiment did not exceed 5 μl.
Key Achievements
- Set of microfluidic devices fabricated by PDMS soft-lithography.
- Characterization of signal transmission range between wildtype MATa and MATx Saccharomyces cerevisiae cells.
- Dynamic characterisation of signal transmission between synthetic MATa and MATx Saccharomyces cerevisiae cells.
- Comparison with mathematical model of signal transmission mechanism and estimation of the activation threshold for different cell concentrations.
- Demonstrated yeast induced blood agglutination on-chip by human antigen A antibody (anti-HuA) displayed on cell surface by Yeast Surface Display.
Introduction
The diffusion processes are slow, and the inertial effects are negligible on micro-scale with low Reynolds number [Angelescu2011]. Hence microfluidics enables complex control of the intercellular microenvironment. Microfluidic experiments in conjunction with live fluorescence microscopy were designed and performed to verify and characterize the signal transmission mechanism in the developed IOD band system, and to verify the developed yeast-induced blood agglutination mechanism.
Soft-lithography
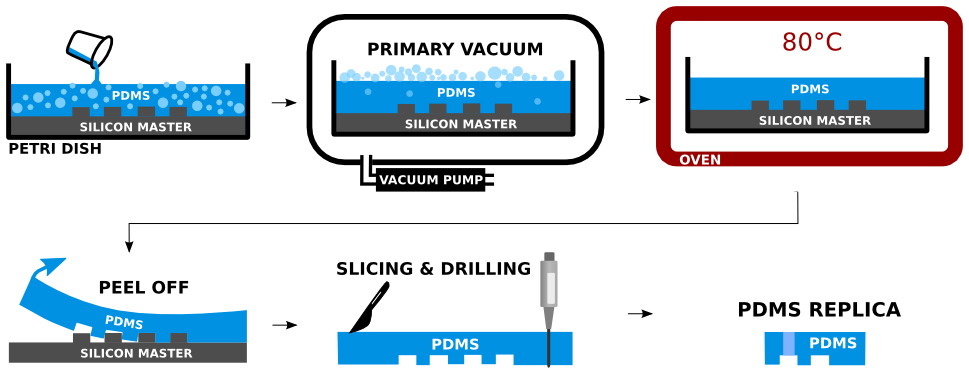
Microfluidic channels were formed using PDMS soft-lithography technology, which has proven biocompatible and readily applicable to available laminar flow cabinets [Fikar2015]. Photomask and silicon master fabrication was outsourced. Fabrication of microfluidic devices was divided into two subsequent steps. In the first step, silicon masters were used for the PDMS molding. In the second step, PDMS molds were bonded to the glass substrates to form encapsulated microfluidic devices using air plasma technology.
PDMS molding
Two-part silicone elastomer Sylgard 184 was used to produce PDMS. The base part was mixed with sufficient amount of curing agent (10:1 ratio). The mixture was centrifuged to remove air bubbles introduced by the mixing. The silicon master was placed in an aluminum foil container and the mixture was poured over. The remaining air bubbles were removed from the PDMS by the sharp tip of a needle. The poured PDMS was maintained in a perfectly horizontal position to assure good planarity and was cured in an oven for 2 hours at 80°C. The PDMS edges were cut off with a sharp tool and the PDMS was peeled off of the silicon master. The PDMS mold was sliced into sections containing individual devices. Inlets and outlets were drilled carefully by biopsy punch of the appropriate diameter at the desired locations of the PDMS replica. Detailed experimental protocol is provided here.
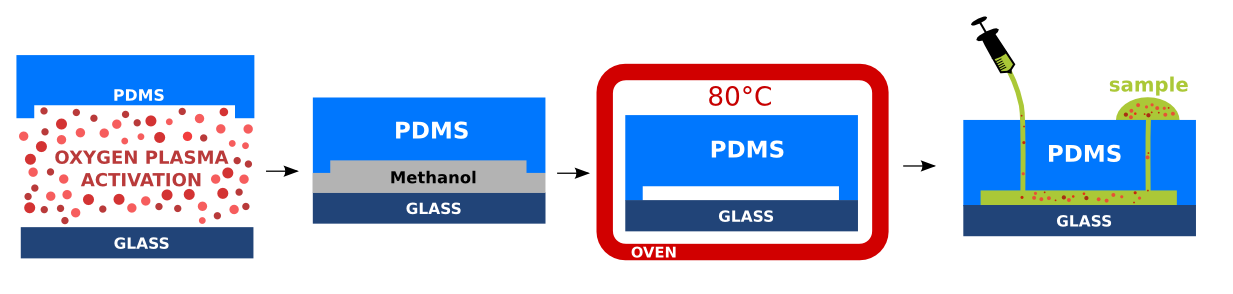
Bonding of PDMS to the glass substrate
Prepared PDMS replicas with imprinted microstructures were cleaned properly with a scotch tape. The PDMS and glass substrate were treated by air plasma for 2.5 minutes. The air plasma affects the PDMS backbone and forms reactive silanol functional groups (Si-OH) enabling formation of permanent irreversible covalent bond of the PDMS to the glass substrate [Wong2009]. In addition, the PDMS treatment with air plasma is beneficial as it avoids non-specific adsorption, decreases cell clogging, and turns the PDMS hydrophilic. The hydrophilicity of PDMS facilitates the future microchannel wetting [Kalio2006]. Immediately after the air plasma treatment, the glass substrate was brought into contact with the PDMS replica and placed in the oven for 5 minutes at 80°C. The bonded devices were stored at room temperature. Inlets and outlets were sealed with scotch tape to avoid contamination. Detailed experimental protocol is provided here.
Experimental setup
Microfluidic experiments were conducted on a platform already established by the Georgiev lab. The laboratory is equipped with a precise microfluidic syringe system (neMESYS Low Pressure Syringe Pumps) and a microscopic station enabling fluorescence imaging and live cell microscopy (Olympus IX83, CellSens software).
Microfluidic experiments
On-chip signal transmission characterization
We developed a new on-chip fluorescence reporter assay system, which allows dynamic detection of GPCR signaling in yeast using live fluorescence microscopy. First, fluorescent reporter proteins are connected to specific functional products signaling activation of the specific G protein–coupled receptor (GPCR). Details on the synthesis and function of reporter plasmids are provided by Module 1. Synthesized reporter plasmids were transformed into specific yeast strains to enable characterization of yeast signaling using the set of orthogonal signals designed and synthesized by Module 2.
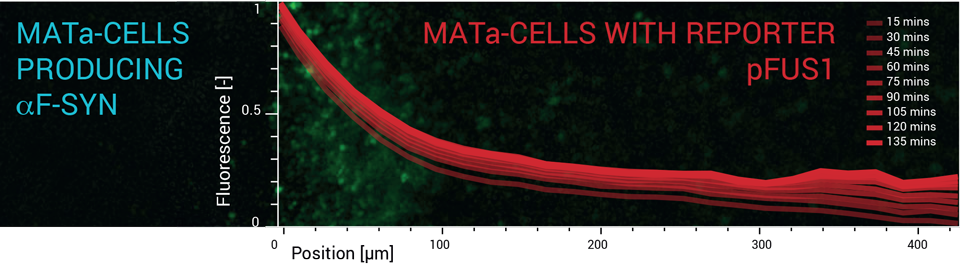
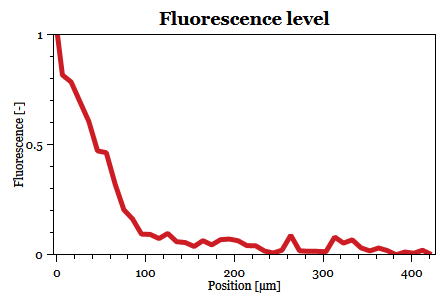
Fabricated microfluidic devices were used to characterize the signal transmission between selected yeast strains. Two cell types were selected for each experiment. One cell type transmits the signal in the form of specific pheromone molecules. And the other cell type produces the green fluorescent protein (GFP) when the signal is received and if the pheromone molecules fit the specific receptor. Both cell types are introduced to the microfluidic device in parallel through separated inlets. Due to the laminar nature of the flow, the individual cell types remain spatially separated inside the microfluidic device. The spatial separation is visible well during the filling of the microfluidic device. The flow is stopped when the filling of the microfluidic device is finished and the rest of the experiment is performed statically. Subsequently, time-lapse fluorescence microscopy is used to dynamically measure levels of GFP expression in the cells that receive the signal.
All experiments were conducted with Saccharomyces cerevisiae cells. Signal transmission was tested between the following types:
- POSITIVE CONTROL: Wildtype MATx ↝ MATa: STE2 receptor from Sacharomyces cerevisiae
- POSITIVE CONTROL: MATa expressing "Candida parapsilosis" alpha-factor; \(\Delta\)Bar ↝ MATa: STE2 receptor from Candida parapsilosis
- NEGATIVE CONTROL: MATa expressing "Candida parapsilosis" alpha-factor; \(\Delta\)Bar ↝ MATa: STE2 receptor from Sacharomyces cerevisiae
Results of the experiment show...
Yeast induced blood agglutination on-chip
Standard agglutination tests are time consuming and require specialized equipment occupying large bench space [Choi2009]. Special microfluidic topologies were designed and fabricated to test yeast-induced agglutination on-chip to lower the time and sample volume requirements. Agglutination tests were performed with synthetic yeast strains inducing blood agglutination by displaying the anti-HuA on its surface. The yeast strains were designed and synthesised by Module 3. The amount of blood necessary for the experiment did not exceed 5 μl.
Personnel
- Martin Cienciala - Microfluidic experiments
- Vaclav Pelisek - Fabrication of microfluidic devices
- Pavel Fikar - Scientific advisor
References
- ↑ Dan E. Angelescu (2011). Highly Integrated Microfluidic Design. Artech House, Norwood.
- ↑ Ieong Wong (2009). Surface molecular property modifications for poly(dimethylsiloxane) (pdms) based microfluidic devices. Microfluid Nanofluid, 7:291–306.
- ↑ Johana Kuncova-Kallio (2006). Pdms and its suitability for analytical microfluidic devices. In Proceedings of the 28th IEEE EMBS Annual International Conference, New York City, USA, 9, IEEE.
- ↑ Sung Hwan Choi (2009). Microinjection molded disposable microfluidic lab-on-a-chip for efficient detection of agglutination. Microsystem Technologies, 15(2):309-316.
- ↑ P. Fikar (2015). SU8 microchannels for live cell dielectrophoresis improvements. In Proceedings of the Design, Test, Integration and Packaging of MEMS/MOEMS Symposium, Montpellier, FR.