Team:Goettingen/Description

About our Project
Our project
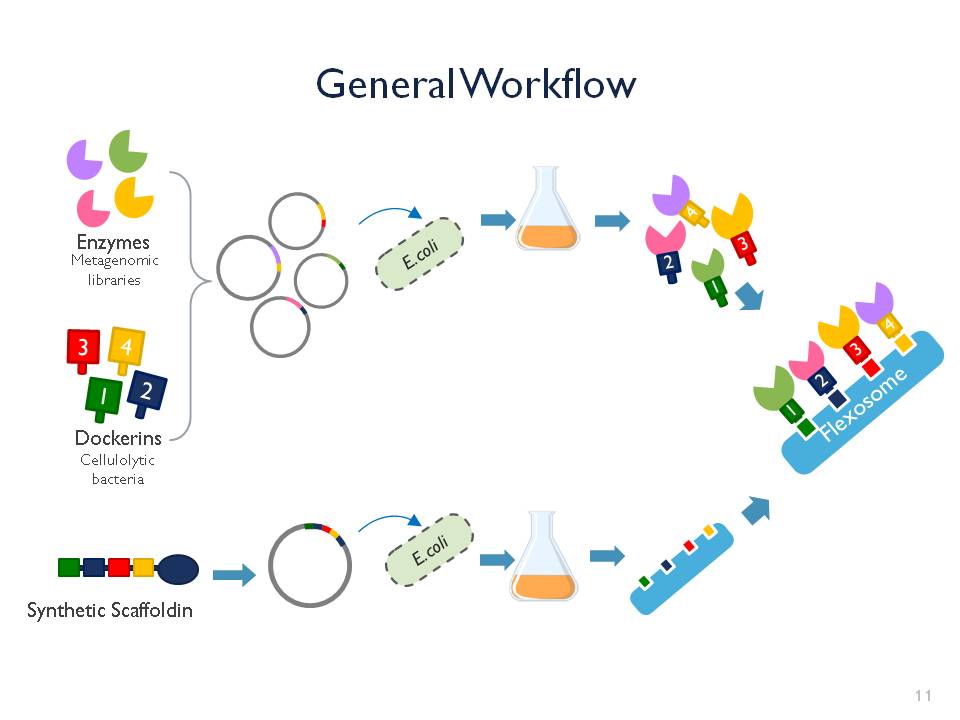
The iGEM team Goettingen 2015 is currently developing the “Flexosome”. It is our enzymatic penknife: a customizable complex for more efficient and synergistic multi-enzymatic processes. It consists of a scaffoldin, dockerins and exchangeable enzymes. The enzymes of interest can be attached to the scaffoldin via the dockerin stations and once attached complete the reactions.The first step
Right now the team is working on a proof of concept. Our first Flexosome will contain phosphatase, esterase and cellulase found in metagenomic libraries along with RFP, a fluorescent reporter gene. Once our E. coli is able to express and produce this functional Flexosome, this will open the door for more complex and specific applications.The inspiration
We studied the natural cellulosome structure, in which different cellulotytic enzymes are assembled into a bigger scaffoldin via dockerin stations, leading to optimized cellulose degradation. The interaction between scaffoldin and dockerins has been proven to be strong and stable, this gave us the ideal platform to design our "Flexosome” device. Traditionally cellulosome engineering is thought for optimization of cellulose degradation, but we don’t have to stop there! A much broader range of enzymes are actively being discovered every year. That is why we intend to combine the scaffoldin protein from cellulolytic bacterium with varying and exchangeable enzymes. The result: a very versatile, stable and innovative tool!The goal
We want to achieve a protein construct which will be able to ensure a high local concentration of enzymes and catalyse multiple enzymatic processes at one site, making the product of one process the substrate of the next on the scaffoldin. Ultimately, the construct will not only increase efficiency of the reactions but also be fully customisable for any kind of enzymatic process.
BioBricks
As our flexosome is built from multiple different domains, we decided that our BioBricks would also consist of these building blocks. Therefore we created BioBricks containing the dockerins alone, as well as in conjunction with a protein. The original plan was to add the enzymes we were using for our proof of principle.
In addition, we decided to use BioBricks from previous and current teams and attach them to our construct, as well as creating composite parts, which would be compatible with our flexosome or any other construct containing the corresponding cohesin domains. We collaborated on this with Aachen and also utilised some previous parts from Uppsala (2011).
In the end we were able to design a basic part containing the Bacteroides cellulolyticus dockerin domain, and a composite part containing the same domain together with eforRed from Team Uppsala 2011. For further information please refer to the registry or our Parts section.
References
General project background
Bayer, E. A., Lamed, R., White, B. A., Flint, H. J. (2008) From Cellulosomes to Cellulosomics. The Chemical Record. Volume 8, Pp. 364 – 377
Blumer-Schuette, S. E., Brown, S. D., Sander, K. B., Bayer, E. A., Kataeva, I., Zurawski, J. V., Conway, J.V., Adams, M. W. W., Kelly, R. M. (2013) Thermophilic lignocellulose deconstruction. FEMS Microbiology Reviews. Volume 38, Issue 3, Pp. 393–448
Chen R., Chen Q., Kim, H., Siu K.-H., Sun, Q., Tsai, S.-L., Chen W. (2014) Biomolecular scaffolds for enhanced signaling and catalytic efficiency. Current Opinion in Biotechnology. Volume 28, Pp. 59 – 68
Ding, S.-Y., Bayer E., A, Steiner D., Shoham, Y. Lamed, R. (2000) A Scaffoldin of the Bacteroides cellulosolvens Cellulosome That Contains 11 Type II Cohesins. Volume 182, Issue 17, Pp. 4915 – 4925
Doi, R. H., Kosugi, A. (2004) Cellulosomes: Plant-Cell-Wall-Degrading Enzyme Complexes. Nature Reviews Microbiology. Volume 2, Pp. 541 – 551
Fontes, C. M.G.A., Gilbert, H. J. (2010) Cellulosomes: Highly Efficient Nanomachines Designed to Deconstruct Plant Cell Wall Complex Carbohydrates. Annual Review of Biochemistry. Volume 79, Pp. 655 – 681
Hirakawa, H. Haga, T., Nagamune, T. (2012) Artificial Protein Complexes for Biocatalysis. Topics in Catalysis. Volume 55, Pp. 1124 –1137
Hyeon, J. E., Jeon, S. D., Han, S. O. (2013) Cellulosome-based, Clostridium-derived multi-functional enzyme complexes for advanced biotechnology tool development: advances and applications. Biotechnology Advances. Volume 31, Pp. 936 – 944
Shoseyov, O., Takagi, M., Goldstein, M., A., Doi R. H. (1992) Primary sequence analysis of Clostridium cellulovorans cellulose binding protein A. Biochemistry. Volume 89, Pp. 3483 – 3487
Xu, Q., Bayer, E. A., Goldman, M., Kenig, R. Shoham, Y. Lamed, R. (2004) Architecture of the Bacteroides cellulosolvens Cellulosome: Description of a Cell Surface-Anchoring Scaffoldin and a Family 48 Cellulase. Journal of Bacteriology. Volume 186, Issue 4, Pp. 968 – 977
Esterase
Montella I. R., Schama R., Valle D. (2012) The classification of esterases: an important gene family involved in insecticide resistance--a review. Volume 107, Issue 4, Pp. 437-449
Nacke, H., Will, C., Herzog, S., Nowka, B., Engelhaupt, M., Daniel, R. (2011) Identification of novel lipolytic genes and gene families by screening of metagenomic libraries derived from soil samples of the German Biodiversity Exploratories. FEMS Microbiology Ecology. Volume 78, Issue 1, Pp. 188-201
Pliego, J., Mateos, J. C., Rodriguez, J., Valero, F., Baeza, M., Femat, R., Camacho, R., Sandoval, G., Herrera-López, E. J. (2015) Monitoring lipase/esterase activity by stopped flow in a sequential injection analysis system using p-nitrophenyl butyrate. Sensors. Volume 15, Issue 2, Pp. 2798-2811
Phosphatase
Castillo Villamizar, G., Nacke, H., Böhning, M., Elisabeth, G., Kaiser, K., Daniel, R. (2014) Novel phosphatases derived from functional metagenomics. 7th International Congress on Biocatalysis. Biocat2014 Hamburg. August 32 –September 4, 2014, P. 87
Greiner, R., Farouk, A.-E. (2007) Purification and Characterization of a Bacterial Phytase Whose Properties Make it Exceptionally Useful as a Feed Supplement. The Protein Journal. Volume 26, Issue 7, Pp. 467-474