Team:Goettingen/Design
Design
Designing the Flexosome
The Flexosome system consists of two sub-units, firstly the scaffoldin subunit consisting of cohesin modules with linker regions between them and secondly enzymatic sub-units consisting of different enzymes linked to dockerins.
In nature, the scaffoldin most often consists of structurally similar cohesins belonging to one family within a given species. The dockerins have strong, specific interactions with their compatible cohesin modules.
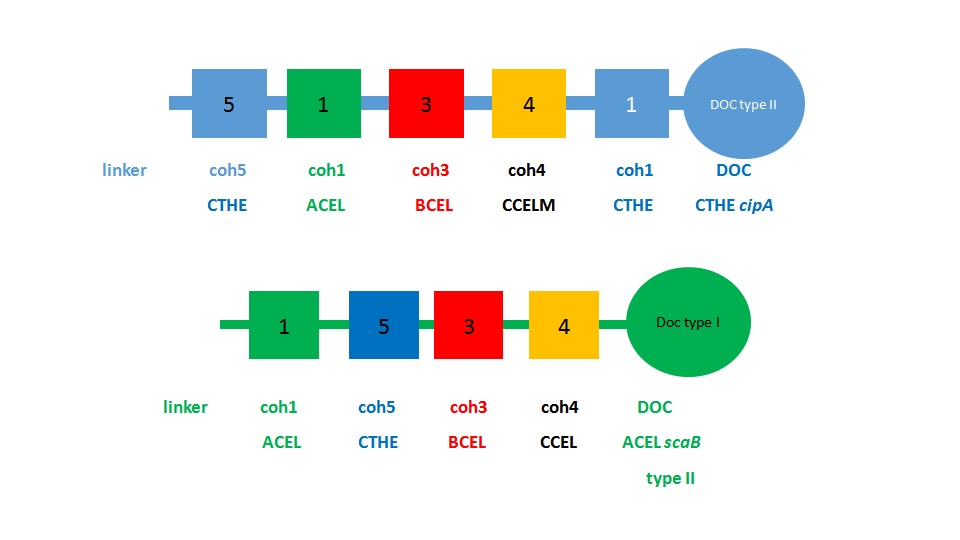
In order to have different functional enzyme-dockerin units attached to the scaffoldin, the cohesin domains had to be synthetically altered by using domains from different cellulosome forming bacteria, namely Acetivibrio cellulolyticus (ACEL), Clostridium cellulolyticum (CCEL), Clostridium thermocellum (CTHE) and Bacteroides cellulosolvens (BCEL)(Fig.1). Dockerins from each organism were chosen for their known interactions with the cohesins that were used. The sequences were obtained from the bacterial genome of the aforementioned bacterial species.
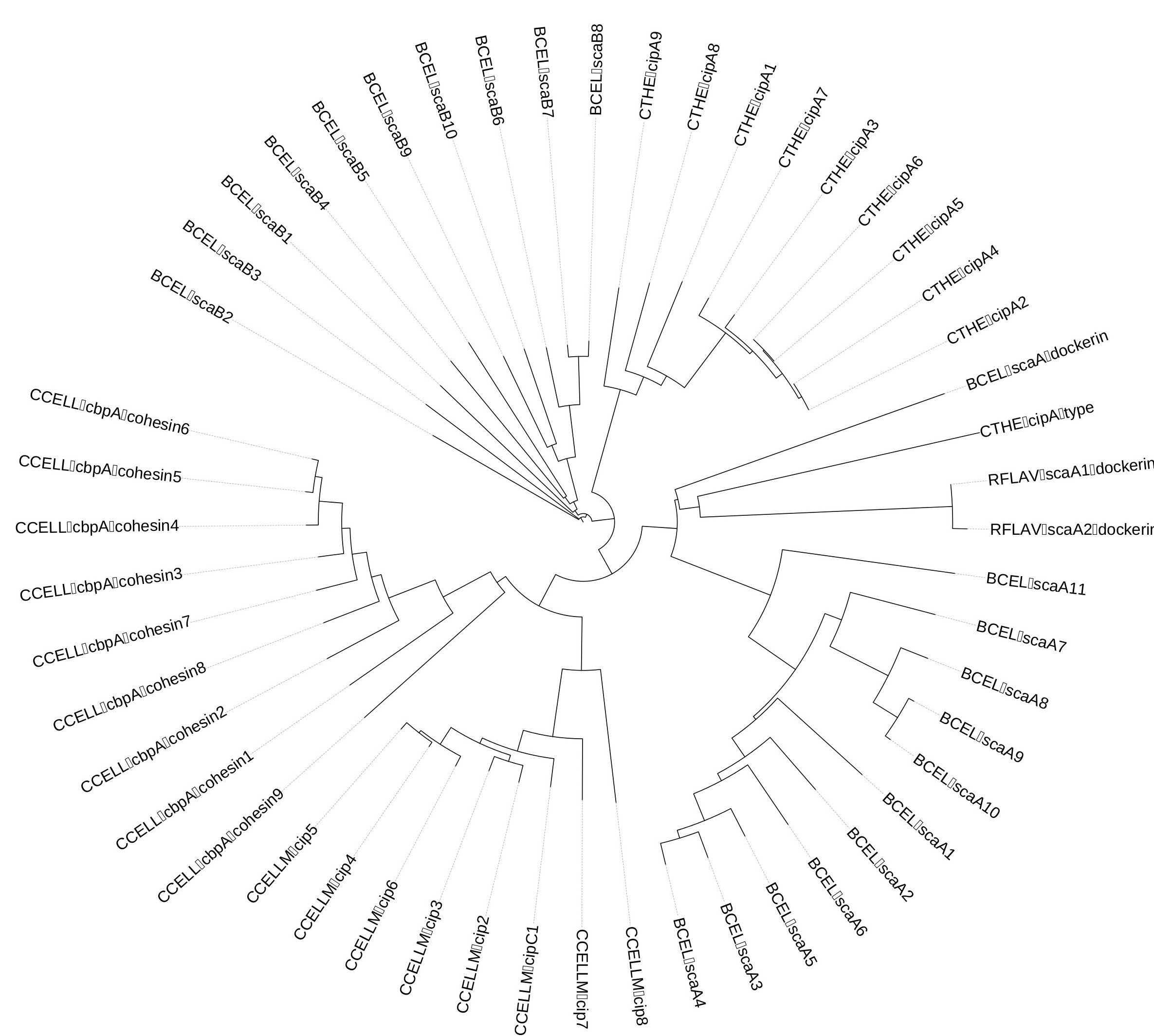
The scaffoldin sequences from Clostridium thermocellum and Acetivibrio cellulolyticus were used as a basis for designing the synthetic scaffoldin. We kept the C-terminal and N-terminal sequences of the scaffoldin molecule unaltered, as we hoped this would maintain the structural stability of the scaffoldins. The sequences of the cohesin modules were pooled and compared for each cohesin family (Fig.2). As they are similar, their consensus sequence was then incorporated into the synthetic scaffoldins. The linker regions between the cohesin modules were left unaltered from the original scaffoldin sequences.
APPLICATIONS
The flexosome does not simply have one application but forms a basis for the enhancement of any enzymatic pathway or combinations, which otherwise have to occur in separate reactions.
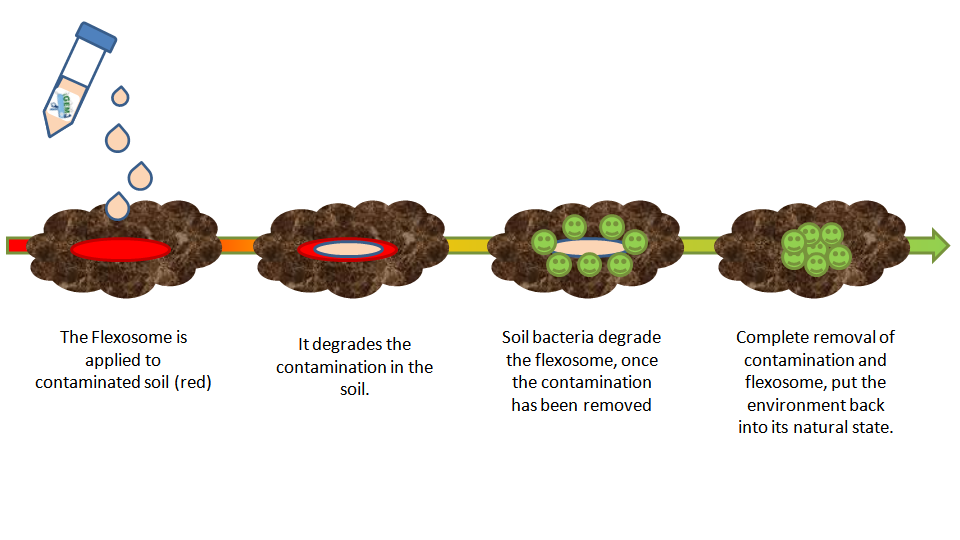
Once past the proof of principle stage, the project could be used to produce complex highly synthetic pharmaceuticals, or at the other end of the spectrum, degrade pharmaceuticals in waste water or contaminations in soil (Fig.3)
Alternatively, the flexosome could be used to optimise the production of biofuels and bioethanols. Particularly its original purpose as cellulosome, could simply be optimised.
Overall our project has great potential to optimise already existing processes in industrial, environmental and pharmaceutical applications.