Team:UCLA/Notebook/Materials/29 May 2015
- Silk fibers from yesterday were soaked overnight in their respective coagulation baths (isopropanol and ethanol)
- Today, we placed the fibers in ddH2O to rinse them
- Took some samples onto microscope slides in order to image
- Left the rest of the fibers on weigh boats to dry off
- The silk is incredibly difficult to handle. We absolutely need a mechanical draw in order to handle the fibers better in the future!
- Fibers were extremely brittle.
- Overview of these fibers
- 15% w/v silk dope
- Spun into 70% v/v ethanol or isopropanol
- Used a 1 mL syringe attached to 0.0005 inch inner diameter PEEK tubing
- extruded at a nominal rate of 6 uL/min on the syringe pump
- After spinning, left in coagulation bath overnight
- Then bathed in water
- Then taken out and left to dry
- Overall, it seems so far that ethanol is better than isopropanol as a coagulation bath. However, until we incorporate a mechanical post-spin draw, we can't say for sure.
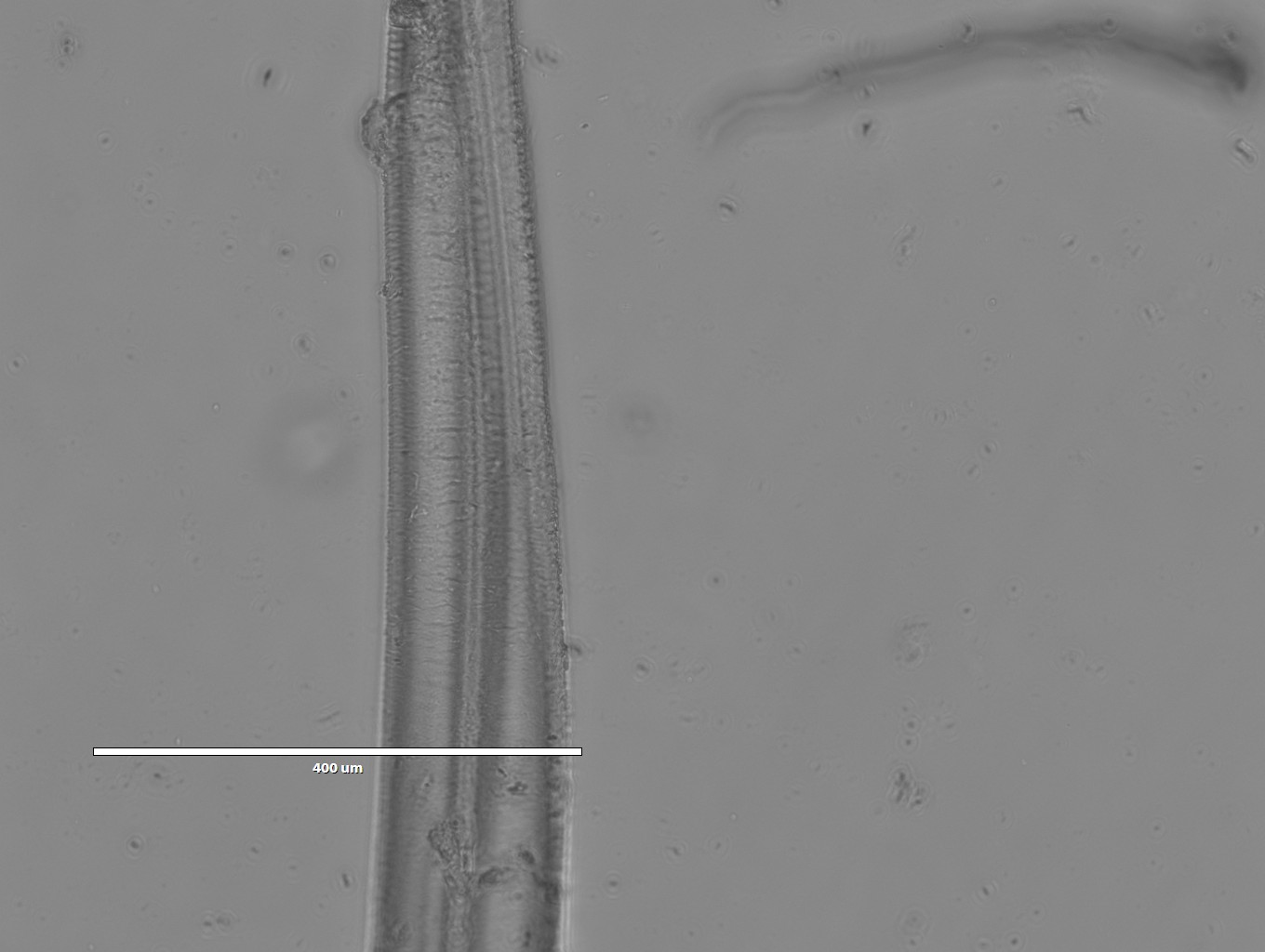
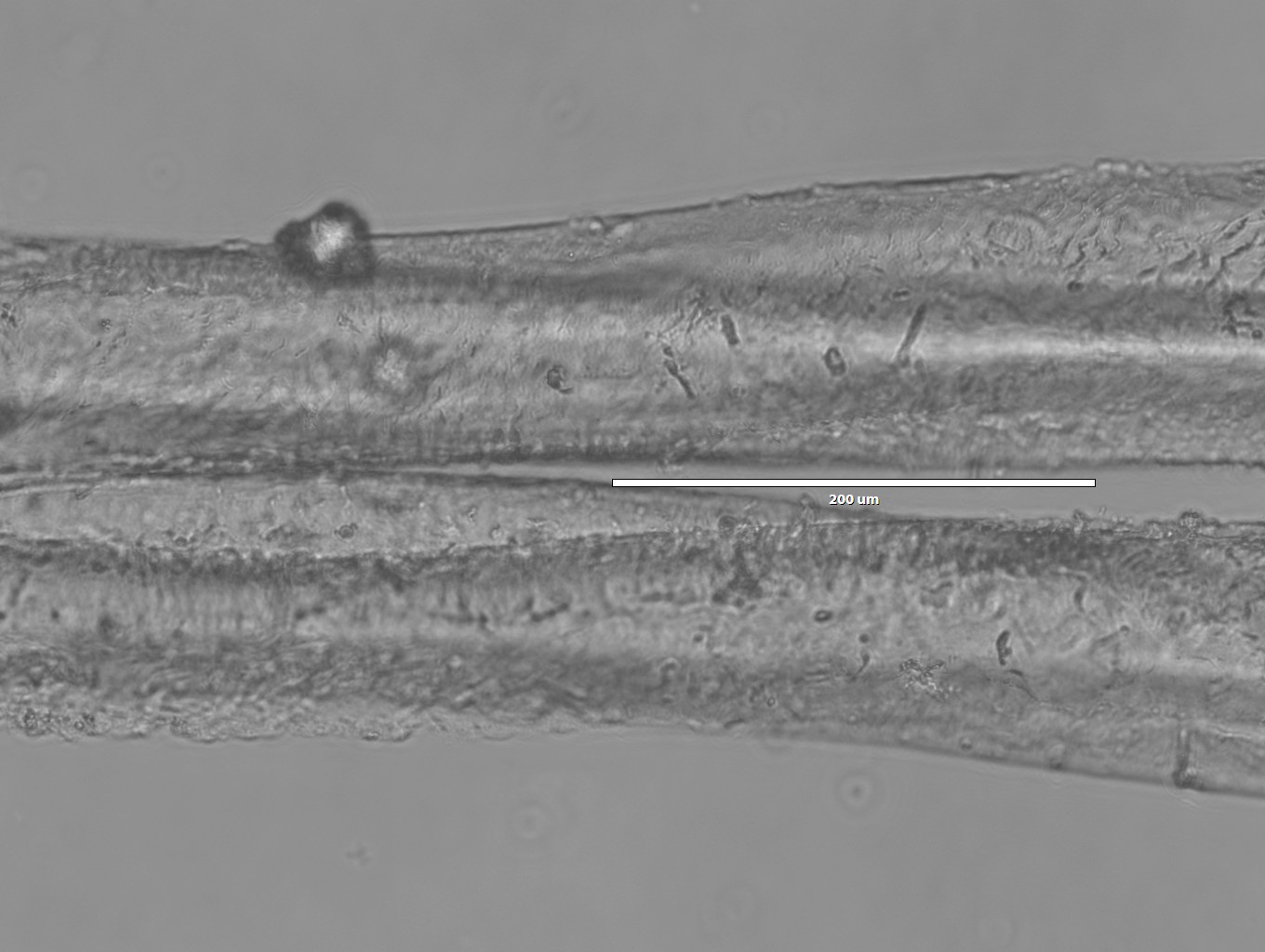
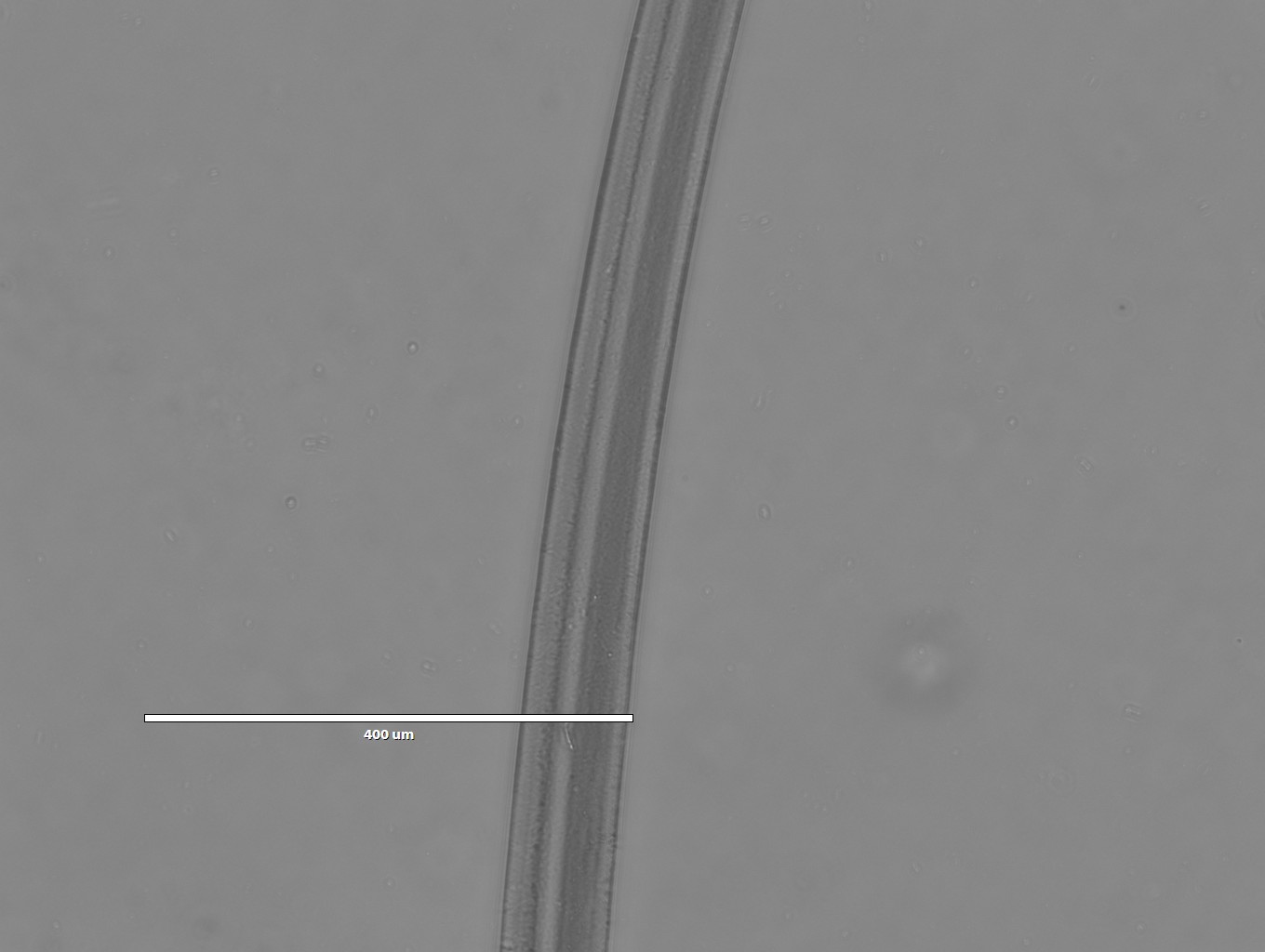
Images of Silk spun into isopropanol
- 4x magnification
- 10x magnification
- 20x magnification
- 40x magnification
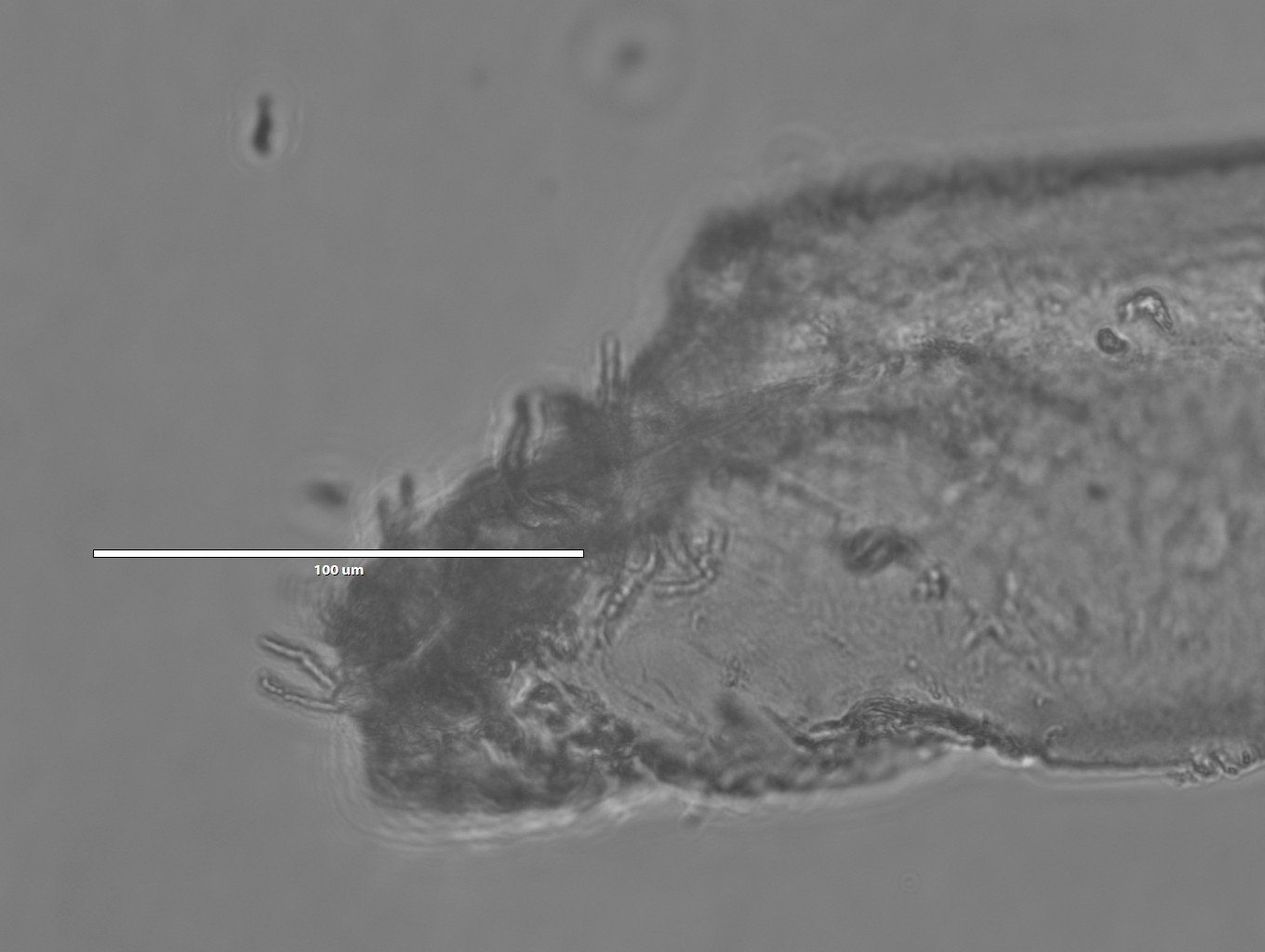
- Those appear to be cracks
- 20x magnification
- Notice the split: we were probably imaging multiple fibers, instead of one. This shows how difficult it is to handle individual fibers while they're still wet!
- 40x magnification
- This is the edge of the fiber where we broke it.
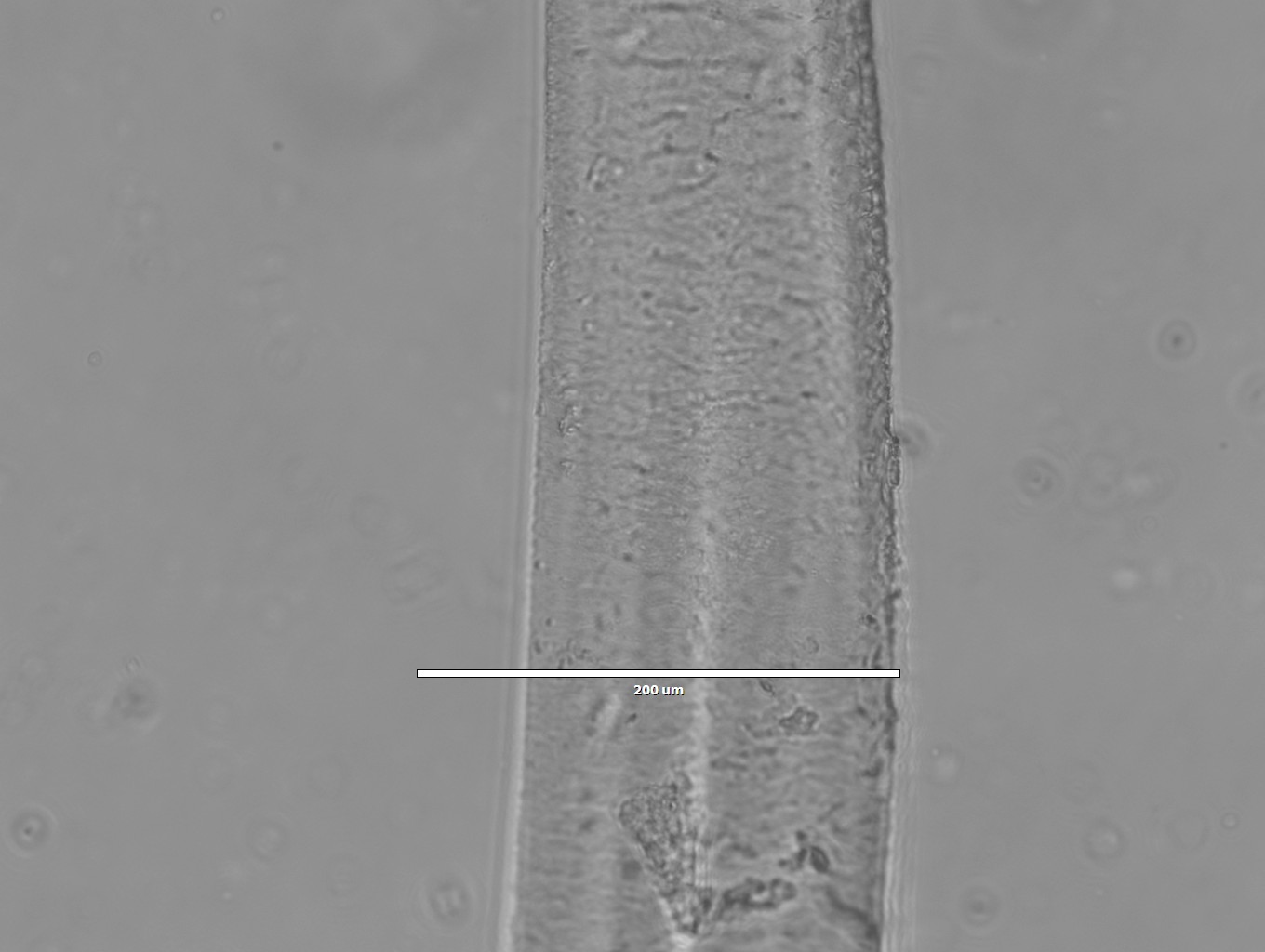
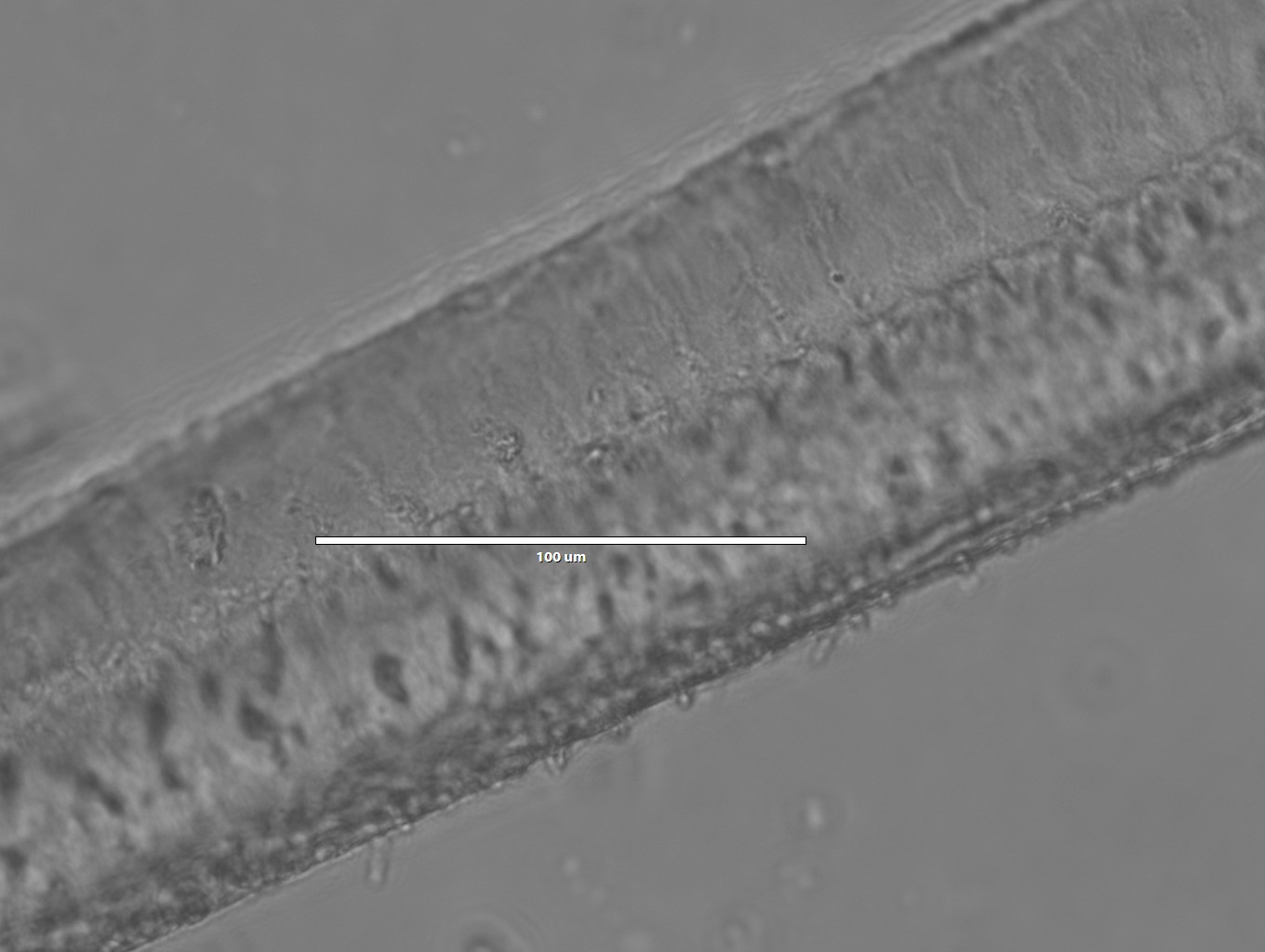
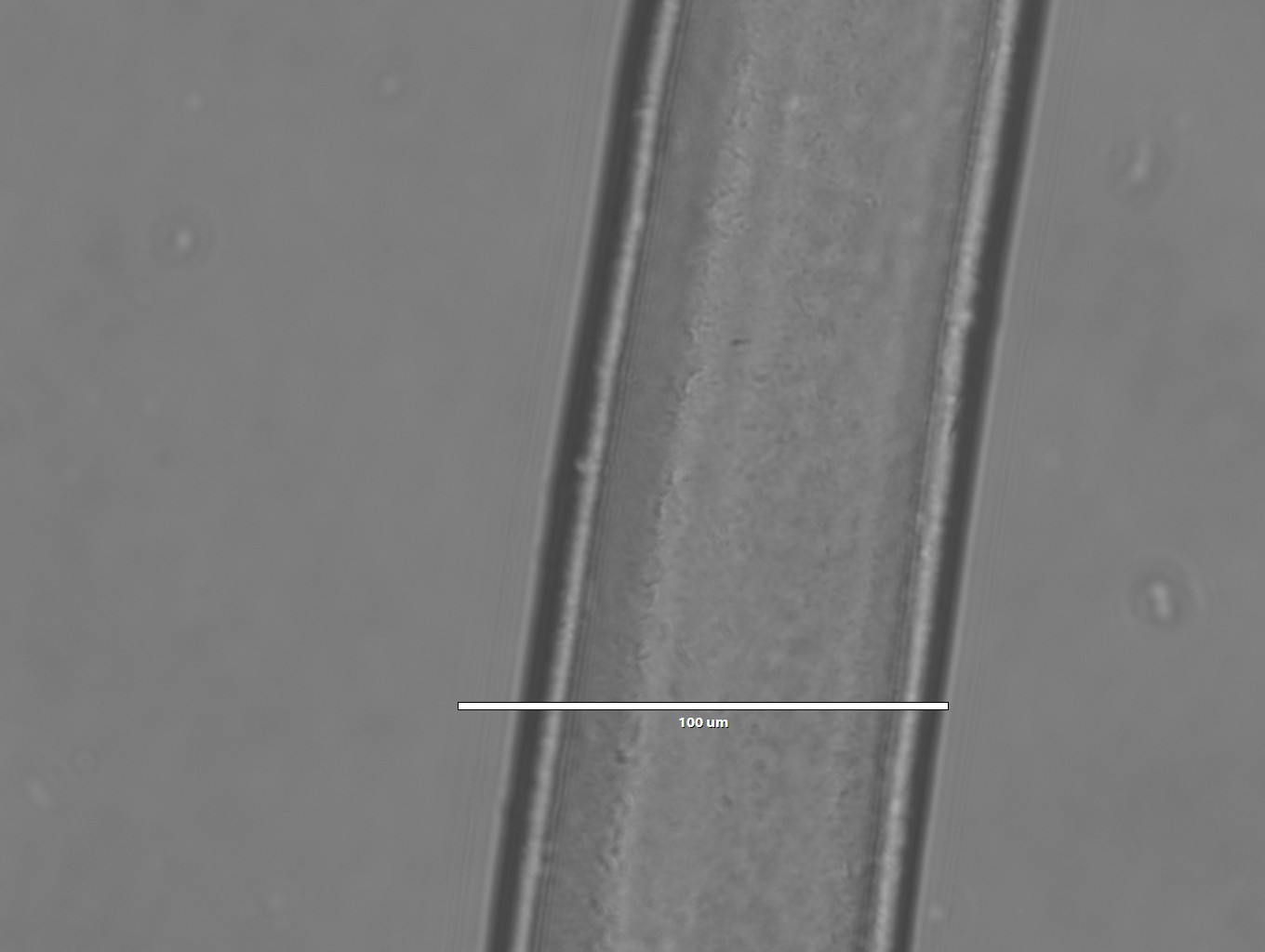
Images of silk spun into ethanol
- 2x magnification
- 10x magnification
- 40x magnification
- Notice how compared to the isopropanol spun fiber, the morphology is much more smooth and uniform for the ethanol fiber. I wonder how these would look under electron microscopy?
Meeting with Julian regarding hydrogels
- making hybrid hydrogels with native bombyx mori silk + Tamura, ABD, etc.
- yields of the recombinant protein
- a few mg yield
- in a buffer
- fairly low concentration
- we'll likely vortex the solution to gelate
- Next week, we'll likely be making negative controls
- have just a silk hydrogel
- measure to see if any protein leaches out from the gel on its own
- we would need to account for this
- make silk + albumin hydrogel
- measure how much albumin leaches out
- make silk hydrogel, put albumin solution on top
- measure how much albumin goes in/ leaches out without the albumin binding domain
- Experimental gels
- make a hydrogel with albumin-binding-domain-silk in it
- pipette a solution of albumin over the top, let it soak for a while into hydrogel. give it time to bind
- remove albumin solution
- pipette some saltwater/buffer solution on top
- at various time points, take aliquots and assay to see how much protein leached out