Team:Czech Republic/Notebook
Notebook
Contents
May
Week 1 (4-10)
Module 2
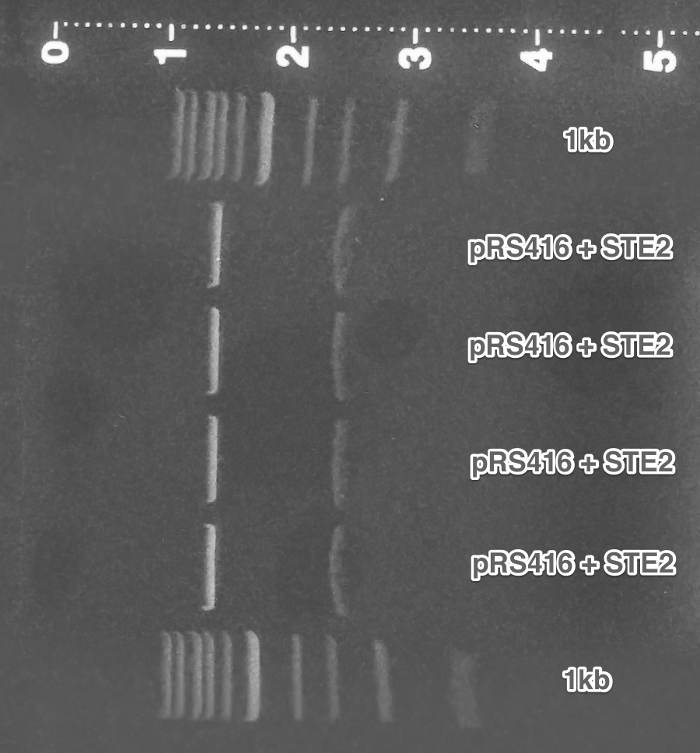
PCR of the ste2 gene from the genome of Saccharomyces cerevisiae 7284 strain (Genome extraction, PCR, Gel electrophoresis).
Cloning of the amplified ste2 into pRSII416 vector (Restriction digest, Ligation, Transformation).
Verification of the pRSII416_ste2 construct (Miniprep, Restriction digest, Gel electrophoresis).
Summary: We successfully obtained ste2 gene from the yeast genome and cloned it into pRSII416 vector.
Module 3
Gel verification of pCT302 yeast display plasmid
Week 2 (11-17)
Module 2
PCR of the incomplete MF(ALPHA)1 locus (see the Design page for details) from the genome of Saccharomyces cerevisiae 7283 strain (Genome extraction, PCR, Gel electrophoresis).
Band-stab PCR of the amplified incomplete MF(ALPHA)1 locus in order to add the actual pheromone and a stop codon.
Cloning of the obtained pheromone into pRSII416 vector (Restriction digest, Ligation, Transformation).
Verification of the pRSII416_sc_alpha construct (Miniprep, Restriction digest, Gel electrophoresis).
Summary: We successfully obtained mating pheromone from Saccharomyces cerevisiae with its secretion tag and cloned it into pRSII416 vector. We also prepared pCUP1 promoter for the cloning upstream of this pheromone.
Module 3
FILLER TEXT
Week 3 (18-24)
Module 2
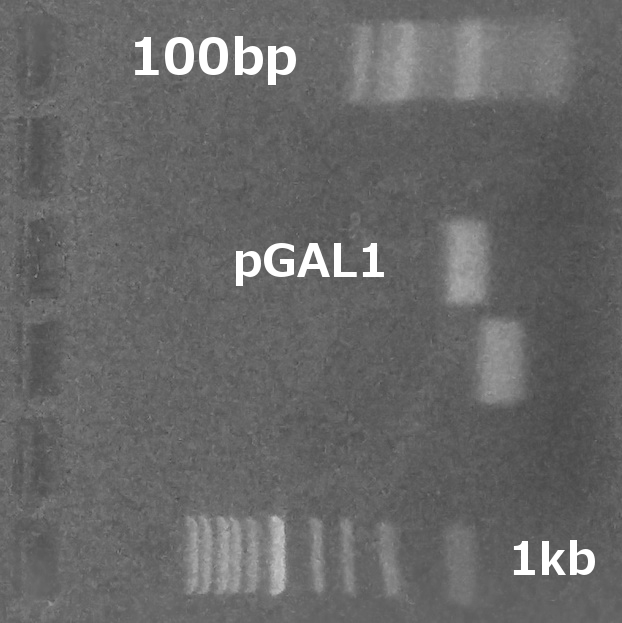
PCR amplification of pGAL1 promoter from the genome of Saccharomyces cerevisiae 7284 strain (PCR, Gel electrophoresis).
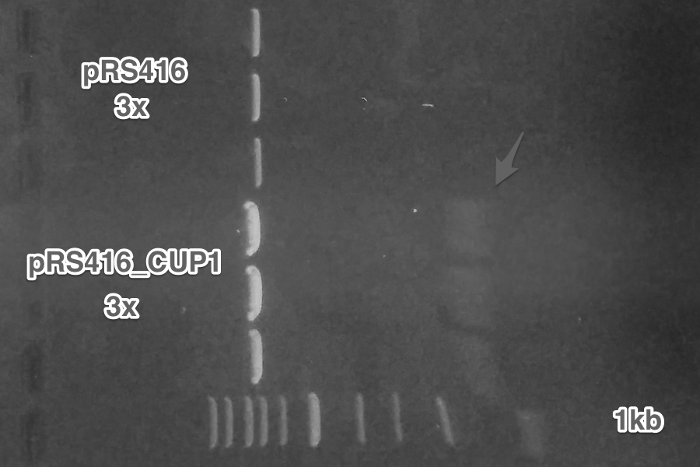
Cloning of the amplified pCUP1 from Week 2 into pRSII416_sc_alpha plasmid (Restriction digest, Ligation, Transformation).
Verification of the pRSII416_pCUP1_sc_alpha construct (Miniprep, Restriction digest, Gel electrophoresis).
Summary: We have successfully obtained plasmid with a mating pheromone under the control of pCUP1 promoter and prepared pGAL1 promoter for the control of ste2 gene.
Module 3
FILLER TEXT
Week 4 (25-31)
Module 3
FILLER TEXT
June
Week 5 (1-07)
Week 6 (08-14)
Week 7 (15-21)
Week 8 (22-28)
Week 9 (29-05)
Module 5
PDMS molding (40μm high silicon masters)
July
Week 10 (06-12)
Week 11 (13-19)
Week 12 (20-26)
Week 13 (27-02)
August
Week 14 (03-09)
Module 3
FILLER TEXT
Week 15 (10-16)
Module 3
FILLER TEXT
Week 16 (17-23)
Module 3
FILLER TEXT
Module 5
Bonding of PDMS to glass (6 microfluidic chips encapsulated on two glass slides)
Week 17 (24-30)
Module 3
FILLER TEXT
Week 18 (31-06)
Module 3
We transformed EBY100 yeast cells with Anti-cMyc, Anti-EpCAM, Anti-HuA and EpCAM inducible yeast display plasmids(Transformation).
Restriction of expression cassette and pRSII 413 and pRSII 415 vectors, subsequent gel purification, ligation and transformation of DH5alpha bacterial cells (Restriction digest, Ligation, Transformation).
Summary: Transformation of EBY100 yeast cells was not successful, no colonies were visible on the solid growing medium. The last attempt of universal expression plasmid (expression cassette on pRSII 413 and pRSII 415 plasmids) construction was not successfull.
Module 5
Signal transmission experiments in microfluidics: MATx + MATa pFUS1-GFP.
September
Week 19 (07-13)
Module 3
PCR amplification of Anti-cMyc, Anti-EpCAM and EpCAM BioBricks from yeast display plasmids (PCR).
Purification of the PCR products, ligation and transformation to DH5alpha bacterial cells (Restriction digest, Ligation, Transformation).
Second attempt of yeast transforamtion with Anti-cMyc, Anti-EpCAM, Anti-HuA and EpCAM inducible yeast display plasmids(Transformation).
Summary: We successfully transformed EBY100 yeast cells with four different types of yeast display plasmid. We also constructed final BioBrick plasmids.
Module 5
Bonding of PDMS to glass (3 microfluidic chips encapsulated on a glass slide)
Signal transmission experiments in microfluidics: MATx + MATa pFUS1-GFP.
Week 20 (14-20)